Definition
- Physiologic state characterised by a systemic impairment in oxygen delivery as a result of reduced tissue perfusion, almost universally mediated by low blood pressure ()
Pathophysiology
- MAP = mean arterial pressure
- CVP = central venous pressure (pressure in the vena cava)
- CO = cardiac output
- Stroke volume is dependent upon preload, contractility and afterload
- SVR = systemic vascular resistance
- Low perfusion pressure must therefore be due to:
- Low preload ⇒
- hypovolaemic (e.g. trauma, GI haemorrhage, severe diarrhoea)
- obstructive ∵ of obstruction to venous return to the LV (e.g. massive PE, pericardial tamponade, tension pneumothorax)
- tachycardic arrythmogenic shock because of short diastolic filling time
- Low contractility ⇒ cardiogenic (e.g. Acute MI, severe heart failure exacerbation, viral myocarditis)
- Low heart rate ⇒
- Bradycardic arrhythmogenic shock
- Low systemic vascular resistance ⇒ distributive (e.g. sepsis, anaphylaxis, spinal cord trauma)
- Other types:
- Toxin-mediated shock (e.g. cyanide and carbon monoxide)
- Low preload ⇒
Classification
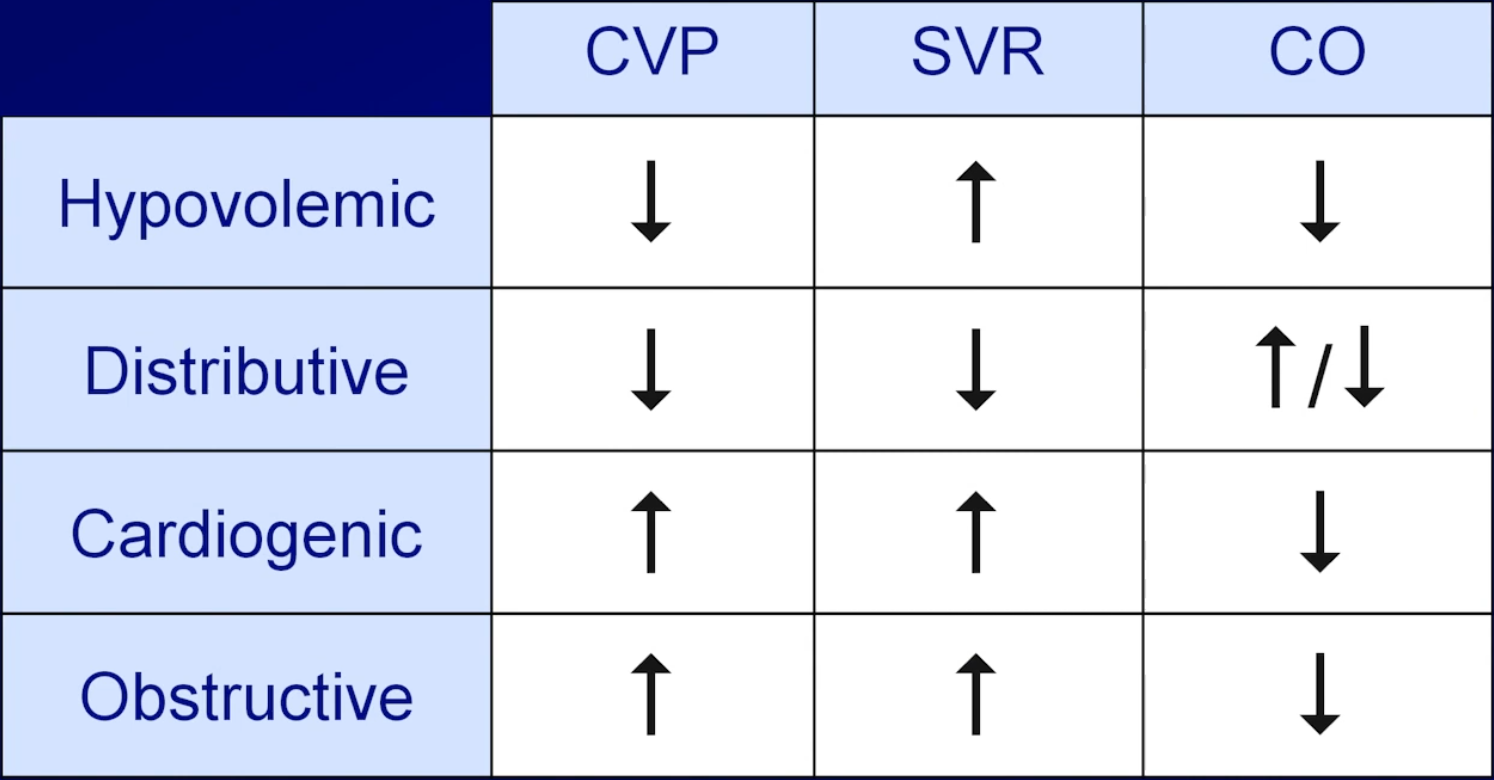
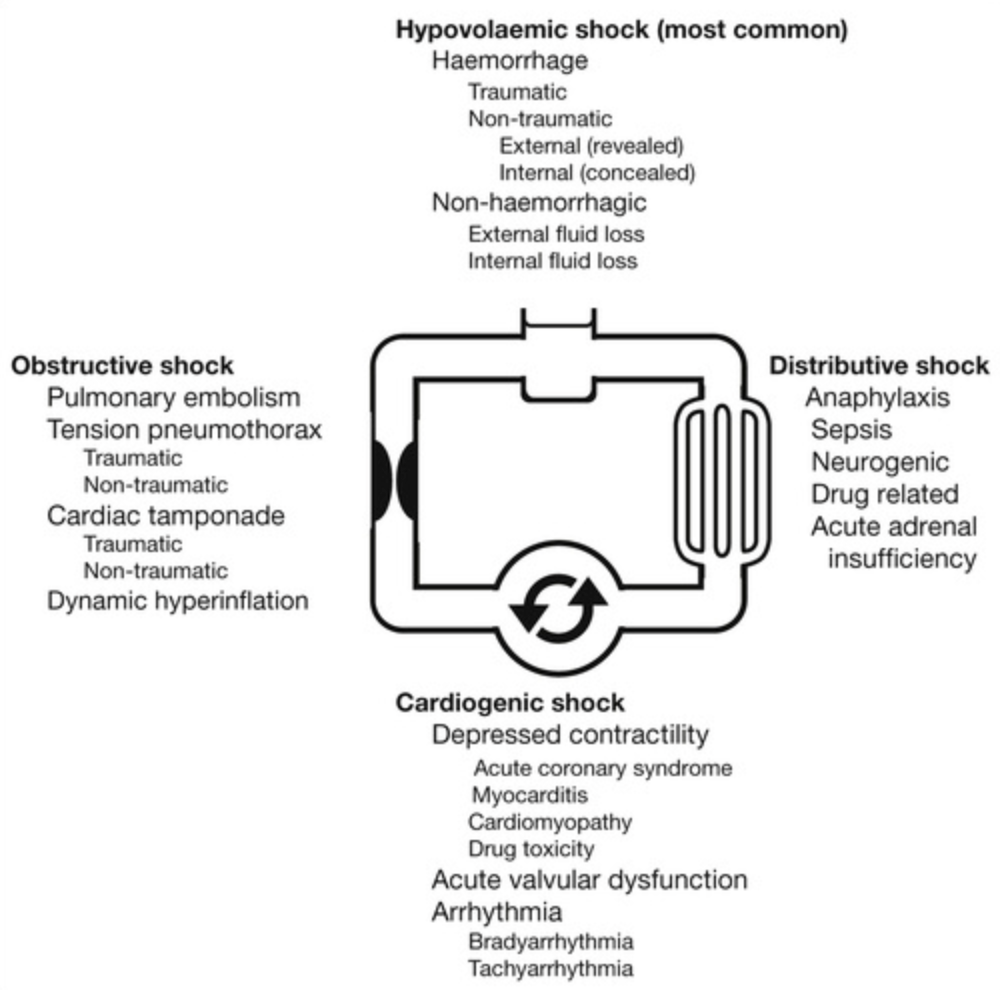
- Cardiogenic shock
- Depressed contractility: ACS, myocarditis, myocardial contusion, cardiomyopathy, drug overdose (e.g. CCB or beta-blocker)
- Acute valvular dysfunction: Papillary muscle or chordae tendinae rupture, infective endocarditis, severe aortic stenosis or mitral stenosis
- Arrhythmia: Tachycardia (e.g. VT, AF, SVT), bradycardia (e.g. heart block)
- Hypovolaemic shock
- Haemorrhagic
- Traumatic: external or internal (e.g. haemothorax, haemoperitoneum, retroperitoneal haemorrhage)
- Non-traumatic: external (e.g. haemoptysis, haematemesis, PV bleeding) or internal (e.g. haemothorax, ruptured AAA, bleeding diathesis)
- Non-haemorrhagic
- External (e.g. GI losses from diarrhoea and vomiting, burns, hyperthermia, high-output fistulae)
- Internal (e.g. bowel obstruction, pancreatitis)
- Haemorrhagic
- Obstructive shock
- Intrinsic to cardiovascular system
- Pulmonary embolism
- Air embolism
- Myxoma
- Amniotic fluid embolism
- Extrinsic to cardiovascular system
- Tension pneumothorax
- Cardiac tamponade
- Abdominal compartment syndrome
- Dynamic hyperinflation: Excessive ventilation with severe bronchospasm (Asthma Exacerbation, COPD)
- Intrinsic to cardiovascular system
- Distributive shock
- Anaphylaxis
- Sepsis
- Neurogenic: Loss of sympathetic tone from high spinal cord trauma or epidural anaesthesia
- Drug related: Vasodilator antihypertensive agents, nitrates, strong analgesics
- Acute adrenal insufficiency: Addison’s disease, discontinuing long-term steroids
- The mnemonic CHOD can be used to remember the above, however a better framework may be PROVED?:
- Cardiogenic (Pump)
- Rhythm abnormalities
- Obstructive
- Hypovolaemia (Volume)
- In the trauma patient consider: SCALPeR: scalp/street, chest, abdomen, long bones, pelvis and retroperitoneum
- Steet refers to external blood loss at the scnes and other pre-hospital haemorrhage
- In the trauma patient consider: SCALPeR: scalp/street, chest, abdomen, long bones, pelvis and retroperitoneum
- Endocrine (these often cause a mixed classification, but its inclusion ensures things like Adrenal Crisis, hypo/hyperthyroidism, Diabetic ketoacidosis, severe acidosis/alkalosis are not missed)
- Distributive
- ?: is it real (check the BP measurement, is the arterial line really in an artery and is the transducer at the correct height)
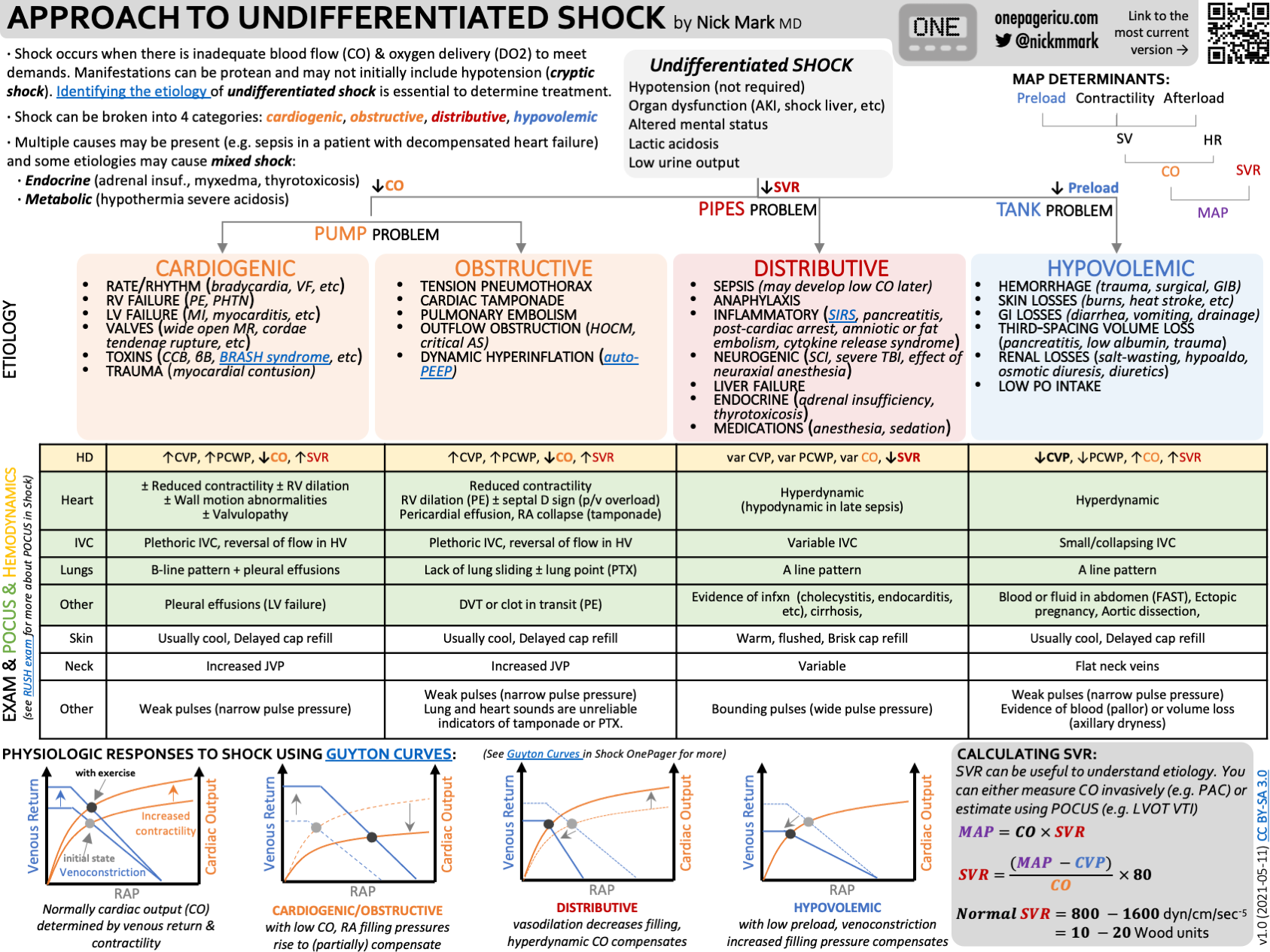
Clinical Features
- Haemodynamics
- Hypotension (MAP < 65 mmHg or significant drop from baseline)
- Elevated shock index (HR/SBP) >0.8
- Urine output < 0.5 mL/kg/hour
- Dark urine
- ↑ troponin
- Skin perfusion
- Cool hands and knees are an early sign of vasoconstriction with reduced cardiac output
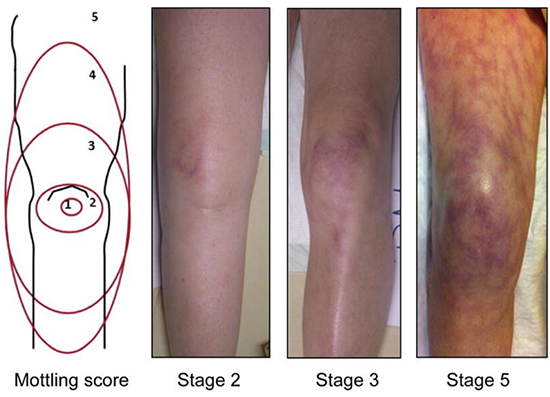
- Mottling
- Suggests active endogenous vasoconstriction, implying that the patient would benefit from ionotropy not vasopressors
- Capillary refill time (>5 seconds)
- Neurological
- Altered mental status (agitation → delirium → solmnolence), generally more of a sign of septic shock
- Respiratory
- ↑ respiratory rate
- ↓ oxygen saturation
- Hepatological
- ↑ bilirubin, AST, ALT
- Nephrology
- ↓ Urine output
- ↑ Creatine
- Haematological
- ↑/↓ platelets
- ↑ INR which can lead to DIC
- ↑ lactate and ↓
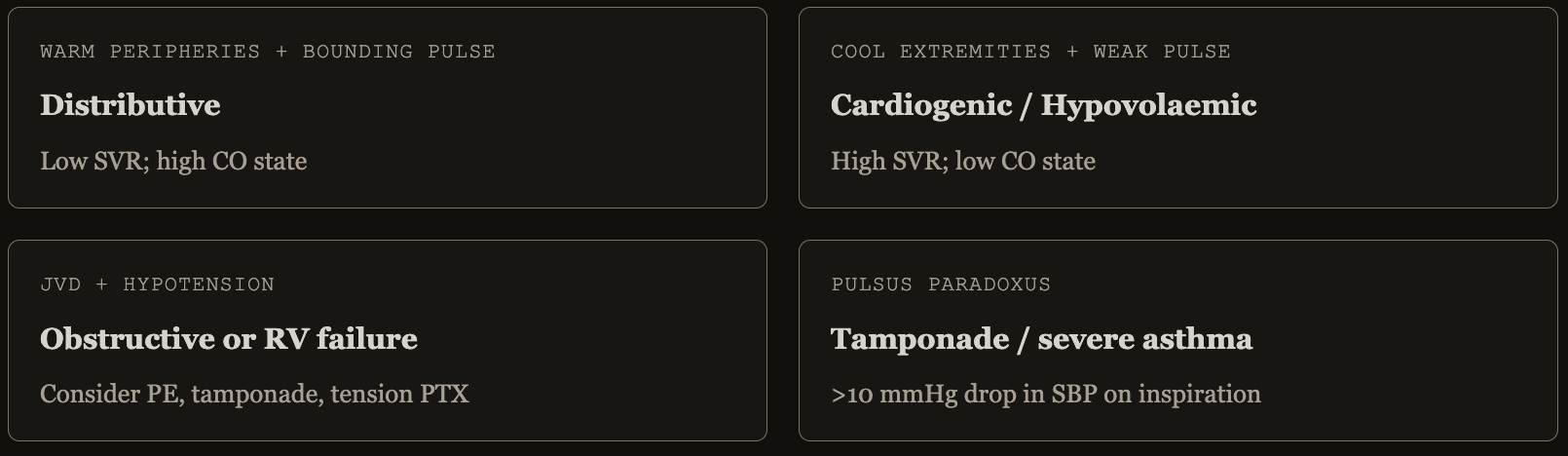
Approach to Undifferentiated Shock
Primary Survey
- Call early for a senior doctor to help
- Assess airway patency
- Exclude airway obstruction and asphyxia
- Start monitoring (pulse oximetry, ECG, BP)
- Assess breathing:
- Oxygenation (saturation probe)
- Ventilation (end tidal CO2, chest auscultation)
- Exclude tension pneumothorax and massive haemothorax
- Assess circulation
- Peripheral and central temperature/capillary refill
- Vital signs (heart rate, blood pressure)
- Equipment check
- Exclude artifactual shock and equipment failure as the cause of shock, e.g. IABP malfunction, ECMO circuit problems, vasopressor infusion drug error.
- Head-to-toe exposure
- Exclude externally obvious haemorrhage
- Exclude anaphylaxis/angioedema
- 12-lead ECG
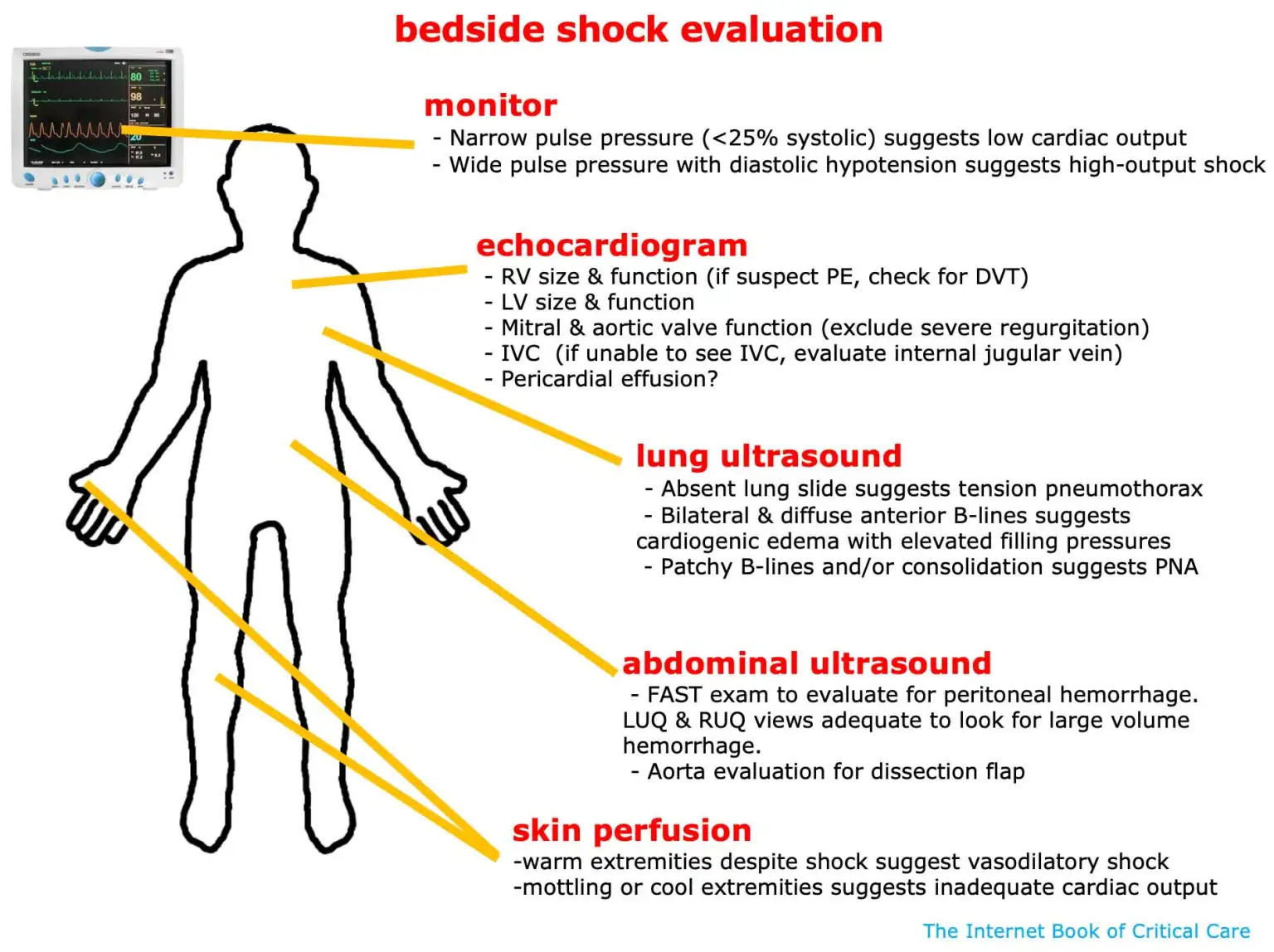
- Point of care TTE: rapid assessment with cardiac echo and RUSH exam
- Exclude cardiac tamponade
- Exclude massive PE
Link to originalOverview of RUSH Exam
- Heart: LV function, RV dilation, pericardial effusion/tamponade
- IVC: size and collapsibility (volume responsiveness)
- Lungs: B-lines (pulmonary oedema), pneumothorax (absence of sliding)
- Abdomen: free fluid (haemoperitoneum, ruptured AAA)
- Aorta: AAA
- Lower limbs: DVT (if PE suspected)
- Establish venous access
- Collect a series of generic laboratory investigations, most importantly an ABG
- Mobile Chest X-ray
- Assess neurology (i.e. spinal injury)
- Exclude neurogenic (“spinal”) shock
- Exclude neurogenic (“spinal”) shock
Empirical Resuscitation
- Ventilate the intubated patient with low-moderate PEEP (0.1cmH2O/kg)
- Commence fluid bolus: 10 ml/kg
- Generally 30 mL/kg within the first 3 hours for sepsis
- Give as a fluid challenge: 250-500mL over 10-15 minutes
- Assess MAP response and dnyamic predictors (PP variations, PLR)
- Balanced crystalloid (hartmann’s or plasmalyte) generally preferred to normal saline unless:
- Hypochloraemic alkalosis (e.g. vomiting, NGT losses)
- Hyperkalaemia
- Traumatic brain injury
- Meningitis
- Albumin
- Albumin 4% reasonable alternative to crystalloid in large-volume resuscitation in septic shock
- Albumin 20% used in hepatic failure/spontaneous bacterial peritonitis, hepatorenal syndrome
- Commence/escalate vasopressor infusion (see below)
- The optimal initial vasopressor is unknown as is the optimal MAP
- In septic shock, it appears the initial pressor of choice is noradrenaline
- The optimal initial vasopressor is unknown as is the optimal MAP
- Antibiotics after peripheral cultures are obtained if sepsis is possible
- Steroids in patients with suspected adrenal crisis
- When in doubt about adrenal insufficiency, a reasonable approach is to give 6 mg dexamethasone and check a cortisol level simultaneously1
Secondary Survey
- Focused history:
- Immediate events preceding the collapse
- Drug administration history
- Recent interventions
- Relevant background history
- Collateral from recently attending staff/family
- Important history points:
- ? chest pain/diaphoresis
- Fevers, rigors, immunosuppression
- Allergen exposure
- Trauma/blood loss
- Known cardiac disease, new dyspnoea
- Chest procedure
- Malignancy (PE)
- Exogenous steroid use or adrenal disease
- Focused examination and investigations
- ECG
- CXR
- ABG with special attention to lactate
- Head to toe examination
- Fluid status (JVP, skin, mucous membranes, heart sounds, lung sounds, CRT, HR, SBP, urine output, IVC and collapsability)
- Sources of sepsis
- Toxidromes
- Abdominal examination, looking for AAA, retroperitoneal haematoma and pancreatitis
- Bedside abdominal and chest ultrasound, looking for collections
- Formal (skilled) TTE, looking for valvular dysfunction, LVOT obstruction, regional wall motion abnormalities and septal defects
- Labs
- FBC
- UEC
- LFT
- Inflammatory markers (CRP and procalcitonin)
- Blood cultures x 2
- Troponin and BNP
- Coagulation studies and D-dimer
- Cortisol level
- TSH and free T4 level
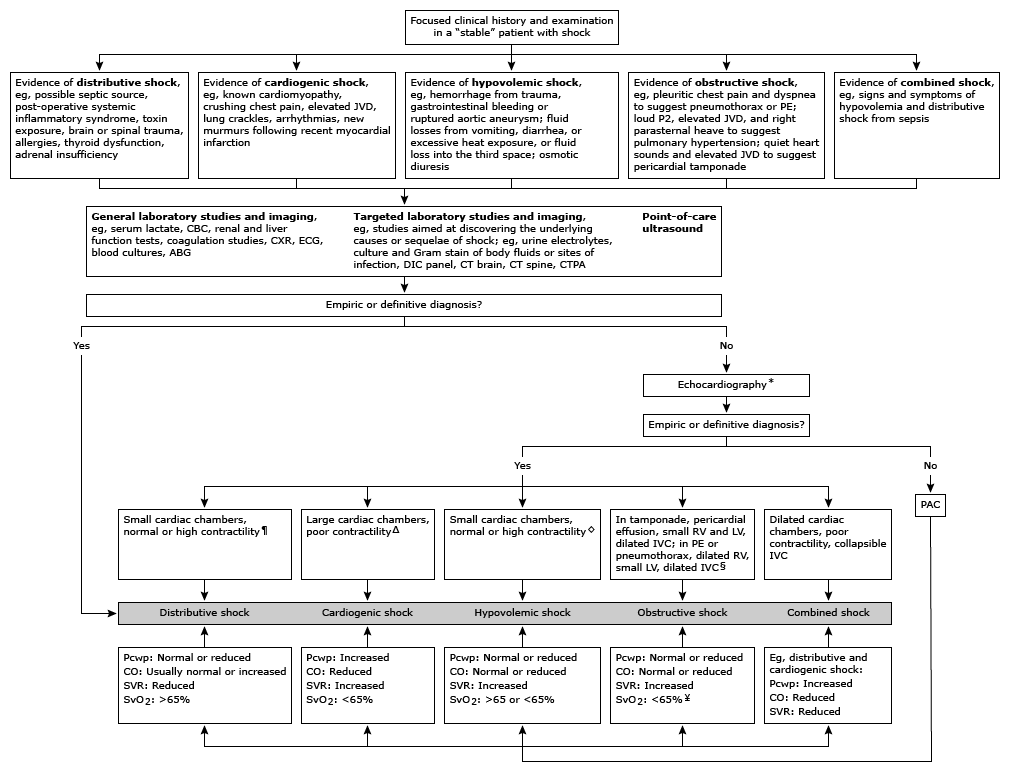
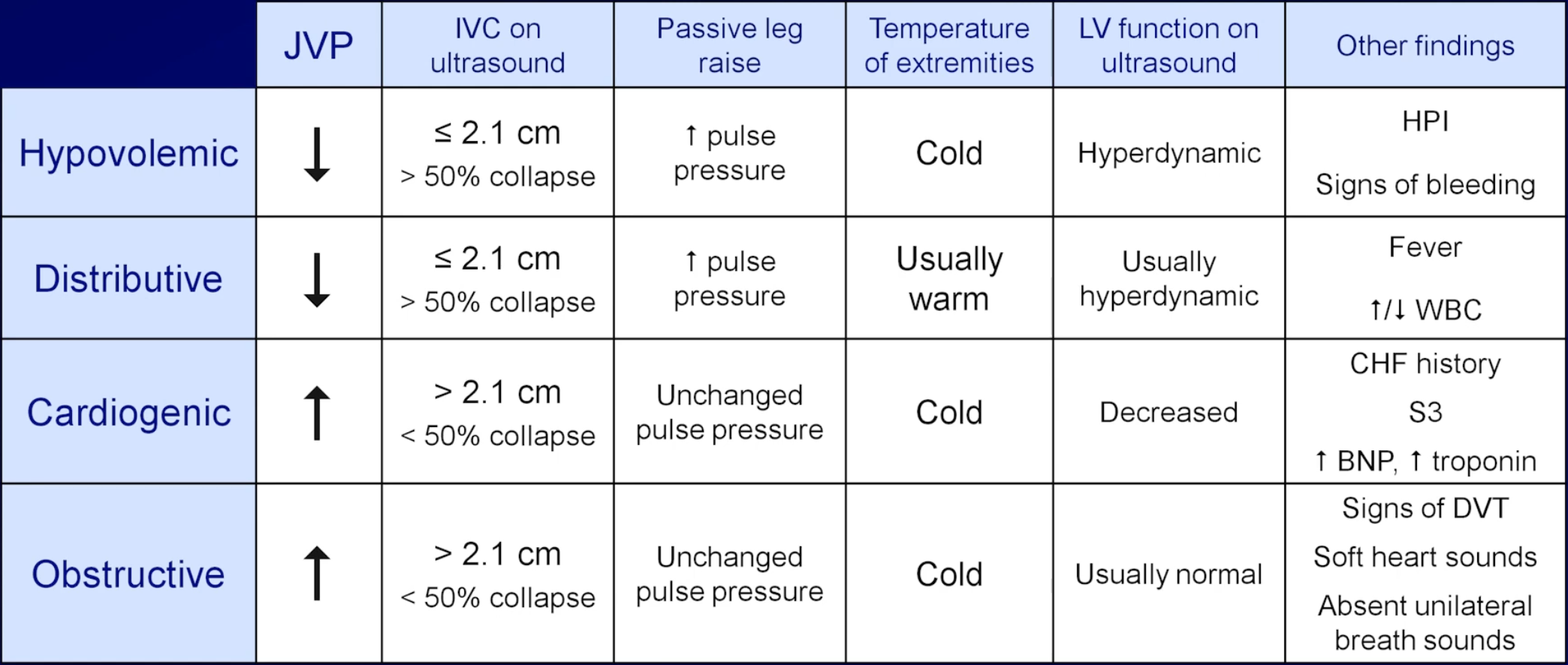
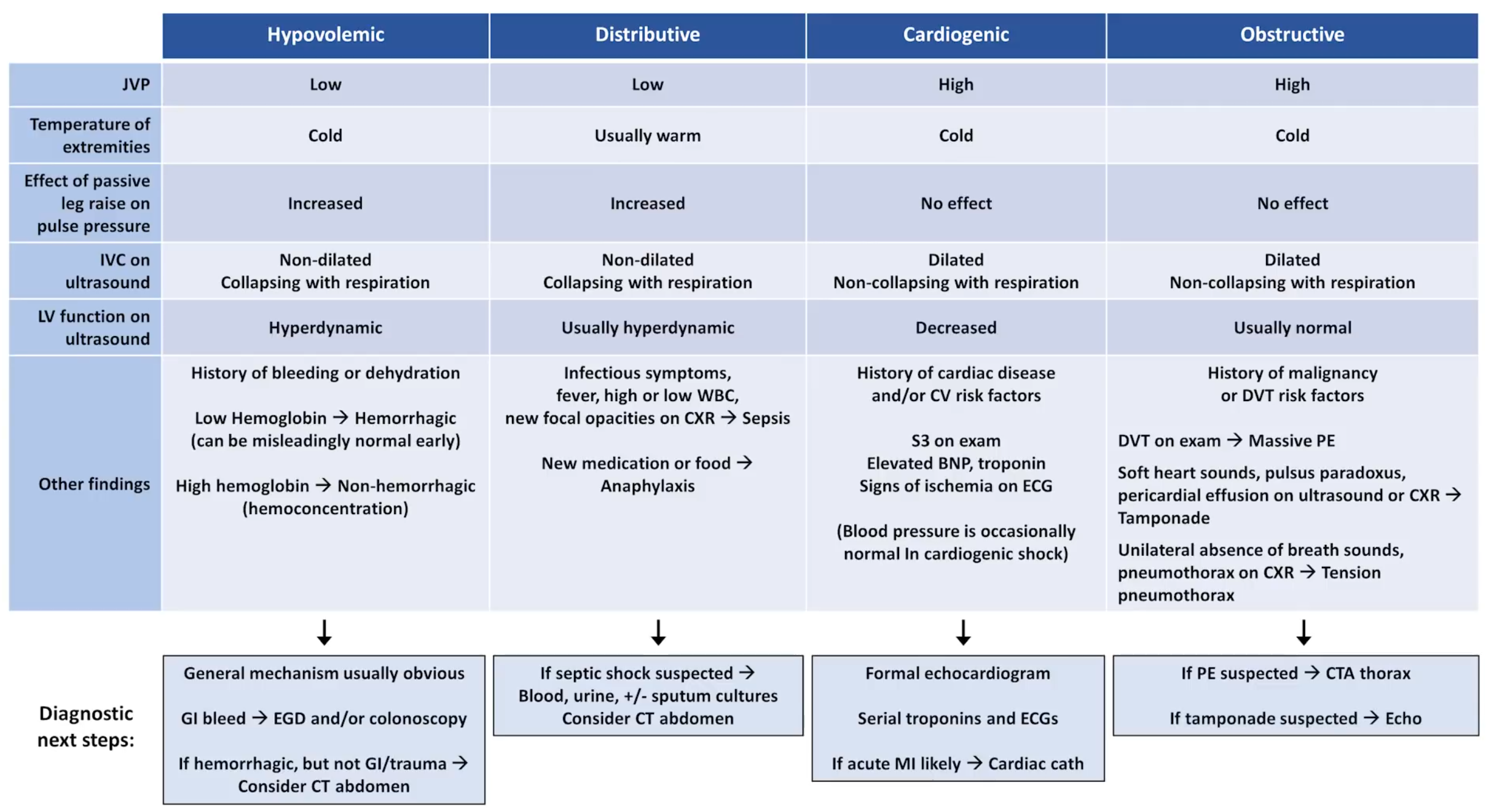
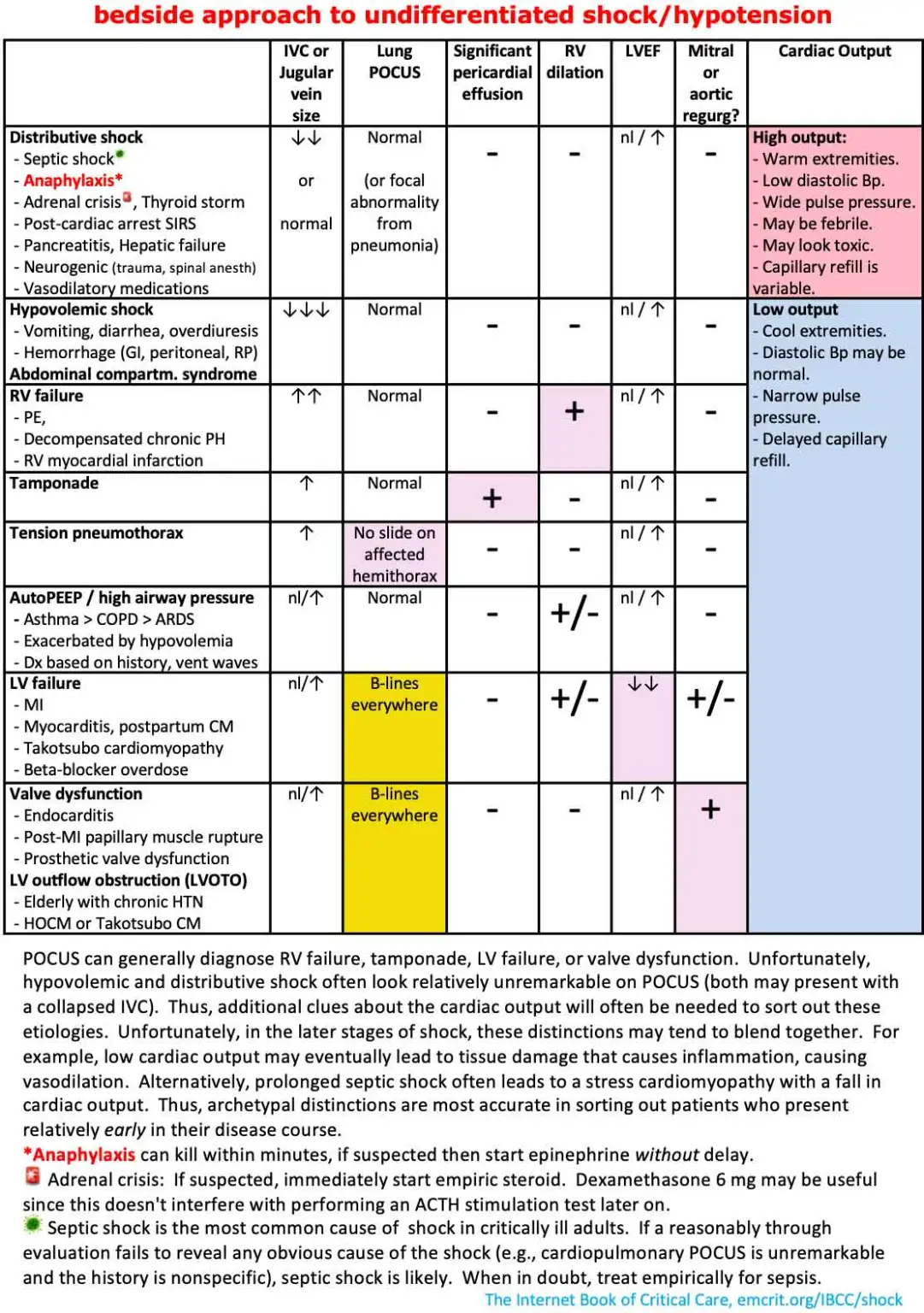
- More than one type of shock may co-exist in the same patient for example:
- Sepsis + hypovolaemia
- Sepsis + sepsis induced cardiomyopathy
Approach to Refractory Shock
- Diagnostic tests:
- Cardiac imaging:
- Repeat POCUS
- Formal echocardiography
- Review CT imaging if available
- Evaluate for unsual forms of shock (e.g. LVOT obstruction)
- Laboratory investigations:
- ABG/VBG and EUCs
- Glucose level
- CMP
- TSH and FT4
- CRP, procalcitonin and blood cultures
- Cardiac imaging:
- Treatment options:
- Optimise MAP target
- Central arterial line (femoral or axillary) as opposed to radial artery
- Reducing MAP target
- Review medications and stop hypotension inducing medications
- Haemodynamically stable analgosedation (e.g. ketamine infusion)
- Discontinue alpha-blockers being used for BPH
- Optimise pre-load
- Reduce PEEP
- Evaluate for auto-PEEP
- Re-evaluate fluid status and consider volume administration if appropriate
- Metabolic optimisation
- Temperature management
- Intravenous calcium in patients with low ionised calcium
- pH optimisation:
- Bicarbonate
- Ventilator management
- Dialysis
- IV thiamine for beriberi
- Steroids may enhance vascular responsiveness to vasopressors
- Thyroid hormone replacement for decompensated hypothyroidism
- Probably treat with T3 to achieve more rapid improvement
- Vasopressor optimisation
- High dose noradrenaline
- Can consider second vasopressor (e.g. terlipressin)
- If mottling present: pivot from vasopressors to inotropes
- Consider right heart catheterisation for refractory or diagnostically unclear shock
- RV optimisation
- Correct all reversible causes of elevated PVR: hypoxia (target SaO₂ >92%), hypercapnia, acidosis, over-distension on the ventilator
- Pulmonary vasodilators: inhaled nitric oxide, nebulised iloprost (prostacyclin analogue)
- Milrinone is the preferred inotrope when there is combined LV + RV failure — it reduces PVR while supporting both ventricles
- Prone positioning reduces PVR in ARDS with cor pulmonale
- Heart rate optimisation
- Atropine
- Isoprenaline
- Pacing
- Mechanical circulatory support
- Invasive aortic balloon pump
- Impella
- VA-ECMO
- Optimise MAP target
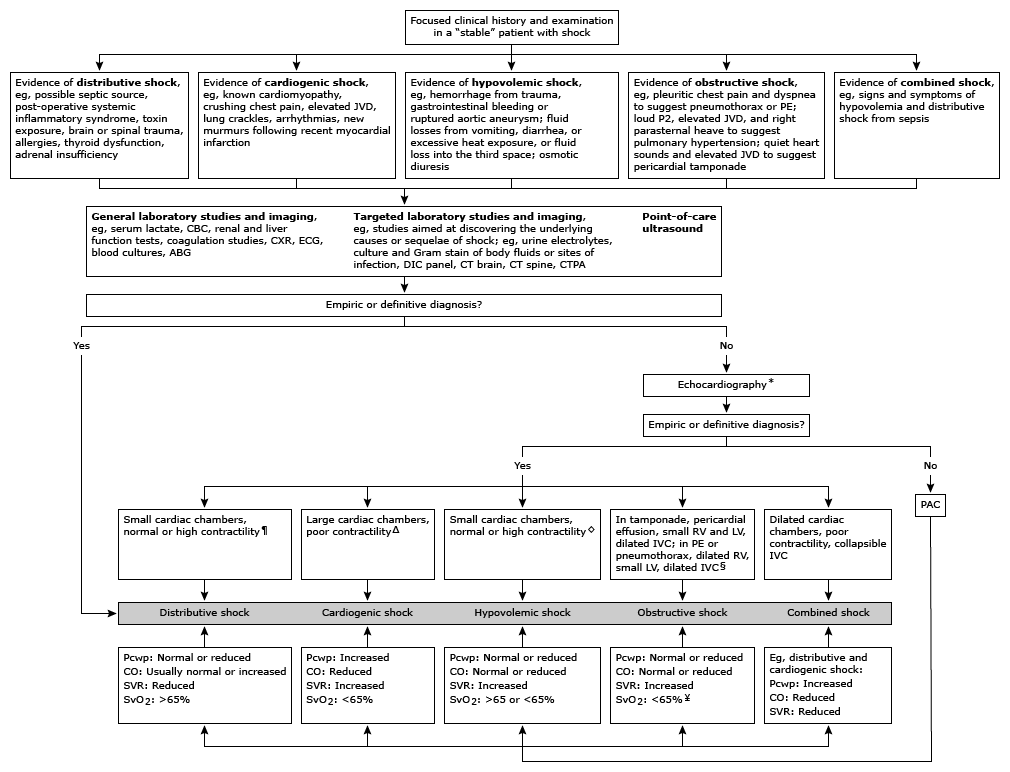
Trauma Related Shock
Vasopressors and Ionotropes
- Start noradrenaline early — don’t wait for large volumes of fluid; fluid overload is harmful
- Peripheral vasopressor use (short-term, large proximal vein) is acceptable to avoid delaying treatment while CVC inserted
- Corticosteroids for vasopressor-refractory septic shock: hydrocortisone 200 mg/day IV (APROCCHSS, ADRENAL trials)
- Titrate to MAP 65 (not higher in most cases — SEPSISPAM trial: no benefit at MAP 80–85)
Link to originalNoradrenaline
0.01-3 mcg/kg/min IV infusion (central line preferred)
First line vasopressor in distributive and most forms of shock
Predominantly agonist with some ⇒ ↑ SVR and mild ↑ HR/CO
Can be run peripherally short-term (forearm or antecubital) at low doses while CVC inserted
Link to originalMetaraminol
Bolus: 0.5-2 mg IV PRN | Infusion: 15-30 mg in 500 mL at 15-60 mL/hr
Predominantly agonist; also causes indirect noradrenaline release
Used widely in peri-operative setting and as bridge vasopressor pre-noradrenaline
Can be given peripherally; suitable in ward or theatre setting
Tachyphylaxis with prolonged use
Reflex bradycardia can occur - treat with atropine
Less titratable than noradrenaline infusion
Link to originalAdrenaline
Anaphylaxis: 0.3-0.5 mg IM (thigh) | Shock infusion: 0.01-1 mcg/kg/min IV
Anaphylaxis: IM adrenaline (Epipen 0.3 mg or ampoule 1:1000) is the drug of first choice
Septic shock: Second agent alongside noradrenaline when additional inotropic support needed; associated with increased lactate that does not reflect true worsening
Cardiogenic shock: Adrenaline has more arrhythmogenic risk than other inotropes
Cardiac arrest: 1 mg IV every alternate loop
Link to originalTerlipressin
Bolus: 0.85–2.5 mg IV bolus q4–6h | Infusion: 1.3–5 mg/24h
- receptor agonist (smooth muscle vasoconstriction)
- Hepatorenal syndrome type 1: terlipressin + albumin is first line
- Variceal bleeding: 2mg IV bolus then 1 mg q4-6h for up to 5 days
- Used in refractory septic shock as noradrenaline-sparing agent (off-label in Australia)
- Risk: digital/skin ischaemia, bradycardia, mesenteric ischaemia — monitor carefully
- Avoid in ischaemic heart disease, peripheral vascular disease
Link to originalDobutamine
2–20 mcg/kg/min IV infusion
- and agonist - positive inotropy, chronotropy; reduces SVR (vasodilatory)
- Used in cardiogenic shock with adequate MAP (often combined with noradrenaline)
- Risk of tachycardia and arrhythmia; may worsen hypotension in true hypovolaemia
Link to originalMilrinone
0.125–0.75 mcg/kg/min IV (load: 25–50 mcg/kg over 10 min, often omitted)
- Used in cardiogenic shock, especially post-cardiac surgery or when β-receptor downregulation limits dobutamine effect
- Useful in pulmonary hypertension (reduces PVR)
- Longer half-life — effects accumulate, harder to titrate; prolonged hypotension if overdosed
- Renally cleared — dose-reduce in AKI
Link to originalPhenylephrine
Bolus 50–200 mcg IV | Infusion 10–300 mcg/min
Pure α₁ agonist — vasoconstriction without inotropic effect
Useful in tachycardia-associated shock where noradrenaline’s β₁ effect is undesirable (e.g., HOCM, AF with fast ventricular rate)
Reflex bradycardia — use cautiously in bradycardic patients
Can worsen cardiogenic shock by increasing afterload without supporting cardiac output
| Situation | First agent | Add / escalate to | Avoid |
|---|---|---|---|
| Undifferentiated shock | Noradrenaline | Fluid trial first; escalate based on 6-step framework | — |
| Septic shock (vasodilatory) | Noradrenaline | Terlipressin (refractory); hydrocortisone; adrenaline if inotrope also needed | Excess fluid, dopamine |
| Cardiogenic shock (LV) | Noradrenaline (MAP support) | Dobutamine or milrinone (inotropy); IABP/Impella/VA-ECMO escalation | Dopamine, excessive adrenaline |
| RV failure | Noradrenaline | Milrinone (reduces PVR + inotrope); adrenaline; inhaled iloprost/NO | Vasodilators, aggressive fluid loading in RV dilation |
| Anaphylaxis | Adrenaline 0.5 mg IM | Adrenaline IV infusion; fluids; glucagon if on β-blockers | Any delay to adrenaline |
| Peri-operative / spinal hypotension | Metaraminol 0.5–2 mg IV bolus | Noradrenaline infusion once CVC placed; phenylephrine if bradycardic | — |
| Bradycardic shock | Atropine → isoprenaline infusion | Temporary transvenous pacing | Phenylephrine, metaraminol (worsen bradycardia) |
| LVOTO / HOCM | Phenylephrine + fluid bolus | Esmolol infusion (rate control) | Inotropes, vasodilators, tachycardia-promoting agents |
| Adrenal crisis | Hydrocortisone 100 mg IV + saline | Noradrenaline if MAP not restored | Delaying hydrocortisone for investigations |
| ![[Pasted image 20260228203914.png | 448]] |
Fluid Responsiveness

- Fluid responsiveness is 10-15% increase in cardiac output in response to a fluid chalenge or pre-load augmentation manoeuvre
- Not the same as needing fluid:
- Is the patient fluid responsive? (Will CO rise with fluid?)
- Does the patient need fluid? (Is tissue perfusion inadequate and likely to improve with more preload?)
- Generally dynamic predictors are better than static predictors (e.g. CVP, PCWP, RAP/LVEDP)
Challenges
Passive leg raise
- Method
- Start with the patient semi-recumbent at 45°
- Raise legs to 45° while simultaneously lowering the torso to flat (or raise legs from flat if already supine)
- This transfers ~300 mL of venous blood from the lower limbs into the central circulation — an autologous, reversible fluid challenge
- Assess CO or a surrogate at baseline and at 60–90 seconds after position change (effect is transient)
- Return patient to original position — the effect is fully reversible
- Positive: ≥10% increase in cardiac output (or arterial pulse pressure as a surrogate)
- Limitations
- Cannot be performed if: raised intracranial pressure, unstable pelvic/femoral fractures, intra-abdominal hypertension (may not transfer adequate volume)
- Requires real-time CO measurement — qualitative BP change alone is insufficient
- Must measure CO response within 60–90 seconds (effect wanes)
Pulse pressure variation
- Positive: PPV > 13% predicts fluid responsiveness
- Limitations: requires all criteria to be met:
- Fully controlled mechanical ventilation (no spontaneous breathing efforts — even small triggered breaths invalidate the result)
- Regular cardiac rhythm (arrhythmias — particularly AF — invalidate the result)
- Tidal volume ≥8 mL/kg ideal body weight (low Vt ventilation, as in lung-protective strategies, reduces cyclic variation even in responsive patients → false negatives)
- No severe RV failure (RV failure independently causes high PPV regardless of volume status)
- Chest closed (open chest post-cardiotomy invalidates)
Mini Fluid Challenge
- Technique: Give 100 mL of crystalloid over 1 minute and assess CO response (via arterial PP or POCUS VTI)
- Positive: ≥6% increase in PP or VTI → likely fluid responsive
Measures
- Best: POCUS VTI (left ventricular outflow tract velocity-time integral) — multiply by LVOT area for CO; serial VTI alone is sufficient for trending
- Acceptable: arterial pulse pressure change (less precise but reasonable)
- Pulse oximetry pleth waveform: some evidence but unreliable in vasoconstriction
- Avoid: relying on BP alone — MAP change is an insensitive surrogate for CO change
IVC collapsibility/distensibility
- Bedside POCUS assessment of the IVC
- In spontaneously breathing patients using the collapsibility index
- Measured in the subxiphoid view
- Collapsibility > 50% suggests fluid responsiveness
- Collapsibility <15-20% suggests volume overload/non-responsiveness
- In mechanically ventilating patients use the distensibility index
- Distensibility >18% predicts fluid responsiveness
Steroids
- Corticosteroids should not be used routinely for patients with septic shock
- the weight of evidence does not support a benefit in terms of patient-orientated outcomes
- ACTH stimulation tests are not useful in this patients
- hydrocortisone 200 mg IV daily is an option for septic shock patients who are refractory to vasopressors (e.g. adults requiring IV noradrenaline at 20-30 micrograms/h) and no other cause found; despite an absence of convincing supportive evidence of benefit
- Corticosteroids should be used in patients who have shock and a specific indication for corticosteroid therapy
- e.g. anaphylaxis, known steroid dependence (e.g. for chronic immunosuppression), known hypoadrenalism, other steroid-responsive conditions like asthma or rheumatoid arthritis
Fluid Replacement in Dehydrated Patients
Fluid replacement in dehydrated patients
- Resuscitate intravascular volume until perfusion is normalised as above with 20 mL/kg boluses of crystalloid (normal saline preferred)
- Calculate fluid losses (generally at least 10% of body weight if patient is dehydrated and hypotensive, i.e. 70kg patient is depleted of 7L of fluid)
- Subtract from this defecit the amount of fluid already given for resuscitation (e.g. if 2L given during resuscitation, a 70kg patient still requires 5L of fluid)
- Replace this amount over the next 24 hours together with maintenance fluid and ongoing losses
- Use 4:2:1 rule, or add 60mL to their weight for hourly fluid requirement ⇒ 110mL/hr ≈ 2.5L/day
- Total fluid to be replaced is 7.5 L
- Replace half in first 8 hours and remainder in next 16 hours
- 3.75L over 8 hours ≈ 450 mL/h
- 3.75L over 16 hours ≈ 230 mL/h
- Monitor adequacy of replacement by perfusion and vital signs, urine output and electrolyte changes
Appendix
| Drug | Alpha-1 | Beta-1 | Beta-2 | Dopamine | Effect on SVR | Effect on HR | Effect on CO | Effect on BP |
|---|---|---|---|---|---|---|---|---|
| Phenylephrine | +++ | 0 | 0 | 0 | ↑↑ | ↓ / ↔ | ↓ | ↑↑ |
| Metaraminol | +++ | + (indirect) | 0 | 0 | ↑↑ | ↔ / ↑ | ↔ / ↑ | ↑↑ |
| Vasopressin (V1 agonist) | 0 | 0 | 0 | 0 | ↑↑ | ↔ | ↓ | ↑ |
| Noradrenaline | +++ | ++ | 0 | 0 | ↑↑ | ↑ | ↔ / ↑ | ↑↑ |
| Adrenaline (Low dose) | + | +++ | ++ | 0 | ↓ | ↑ | ↑ | ↔ / ↑ |
| Adrenaline (High dose) | ++ | +++ | ++ | 0 | ↔ / ↑ | ↑ | ↑ | ↑↑ |
| Dopamine (Low dose)* | 0 | + | 0 | ++ | ↔ | ↑ | ↑ | ↑ |
| Dopamine (Moderate dose)* | + | ++ | 0 | ++ | ↑ | ↑ | ↑ | ↑↑ |
| Dopamine (High dose)* | ++ | ++ | 0 | ++ | ↑↑ | ↑ | ↔ / ↑ | ↑↑ |
| Dobutamine | 0 / + | +++ | ++ | 0 | ↓ | ↑ | ↑↑ | ↓ / ↔ / ↑ |
| Isoprenaline | 0 | +++ | +++ | 0 | ↓ | ↑↑ | ↑↑ | ↓ / ↔ |
| Milrinone (PDE inhibitor) | 0 | 0 | 0 | 0 | ↓ | ↔ / ↑ | ↑↑ | ↓ / ↔ / ↑ |
Sources
- Youtube Videos
- LITFL:
- Deranged Physiology
- UpToDate: Evaluation of and initial approach to the adult patient with undifferentiated hypotension and shock
- Undifferentiated Shock — ICU One Pager
- Vasopressors — ICU One Pager
- Fluid Responsiveness - could this hemodynamically unstable patient respond to IV fluids? — ICU One Pager
Footnotes
-
Dexamethasone does not interfere with the cortisol level allowing one to perform an ACTH stimulation test later if indicated ↩