Presentation
- Develops over minutes with severe breathlnessness, coughing frothy pink sputum, orthopnoea and PND
- Patient is typically distressed, unable to lie flat and usually peripherally shut down and clammy
- Examination findings include tachypnoea, tachycardia with widespread basal crackles and possibly expiratory wheeze (‘cardiac asthma’)
- Patients are often hypertensive, often referred to as sympathetic crashing acute pulmonary oedema (SCAPO). These patients are profoundly vasoconstricted with significantly elevated afterload
Diagnostic Dilemma
Any form of severe respiratory failure may lead to distress and hypertension, therefore don’t assume that respiratory failure and hypertension implies the diagnosis.
Aetiologies
| Cardiogenic | Non-Cardiogenic |
|---|---|
| Excessive LV afterload (i.e. SCAPO) (e.g. severe hypertension, aortic stenosis, HOCM) | Increased capillary permeability (e.g. ARDS, reperfusion following relief of embolic occlusion, near drowning) |
| Excessive LV preload (e.g. fluid overload, rapid fluid bolus) | Neurogenic pulmonary oedema |
| Excessive left atrial afterload (e.g. mitral stenosis, mitral thrombosis, mitral valve prolapse, atrial myxoma) | Drug-induced pulmonary oedema (e.g. opiate induced, salicylate overdose) |
| Poor contractility (e.g. cardiomyopathy, drug effect (e.g. beta-blockers), metabolic disease, myocarditis) | Raised pulmonary arterial pressure (e.g. massive PE, pulmonary veno-occlusive disease, air embolism) |
| Ineffective contractility (e.g. mitral regurgitation, Takotsubo cardiomyopathy) | Negative pressure pulmonary oedema (e.g. airway obstruction, after drainage of a large pleural effusion) |
- Often underlying risk factors are present concurrently with the acute triggers in the table above:
- Chronic left ventricular failure (especially diastolic heart failure)
- History of hypertension (especially labile hypertension)
- Renal artery stenosis predisposes acute pulmonary oedema (Pickering syndrome)
Investigations
ABG results
- Initially in APO patients are sympathetically driven and hyperventilating resulting in a picture of respiratory alkalosis (↑ pH ↓ )
- As pulmonary oedema worsens, alveolar flooding and fatigue causes hypoventilation causing respiratory acidosis (↓ pH ↑ )
- Occassionally mixed picture acidosis (metabolic + respiratory acidosis) can arise (e.g. acute pulmonary oedema after an acute myocardial infarct), caused by poor cardiac circulation (causing a lactic acidosis – metabolic acidosis) with concurrent poor alveolar ventilation (due to pulmonary oedema) – causing retention and a concomitant respiratory acidosis
- See: Myocardial infarction with acute pulmonary oedema for an example
- Mixed picture respiratory alkalosis and metabolic alkalosis (from aggressive diuretic therapy) present
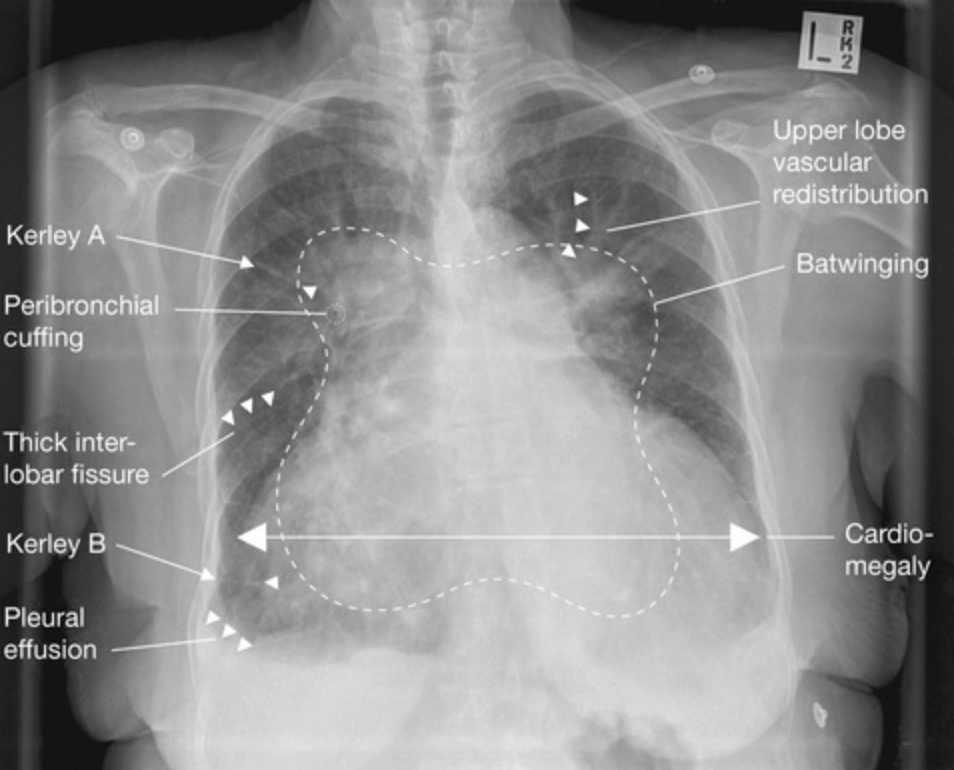
Chest-Xray
- Alveolar oedema (‘batwing’ appearance)
- Kerly B lines (1-2cm horizontal, peripheral engorged subpleural lymphatics) and septal lines
- Cardiomegaly
- Upper love diversion
- Pleural effusion
- Additionally to ABCDE features above; it may show a precipitating cause such as a pneumonia
- Progression
- Interstitial pulmonary oedema develops first
- Peribronchial cuffing
- Thickening of interlobar fissures
- Bat wing oedema is alveolar oedema in a non-gravity-dependent distribution; usually seen in hyper-acute heart failure (e.g. acute mitral incompetence due to papillary muscle rupture)
Point of Care Ultrasound1
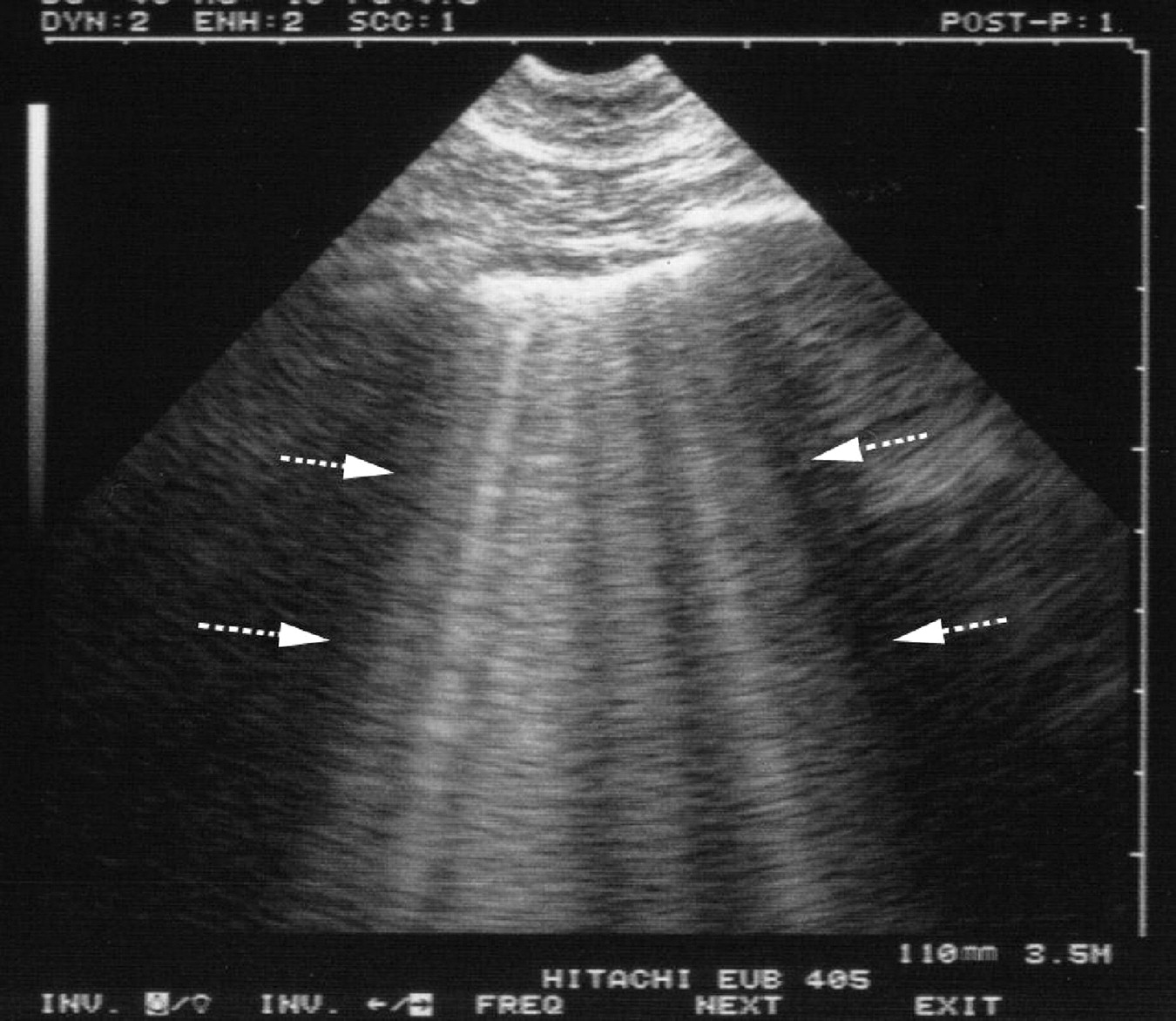
- B lines seen on lung ultrasound
- The presence of ≥3 B-lines per rib space suggests the presence of interstitial pulmonary fluid
- Cardiogenic pulmonary oedema generates a pattern of diffuse B-lines throughout the chest with thin pleural interfaces
- Non-cardiogenic pulmonary oedema shows a patchy distribution of B-lines anda. thickened pleural interface
- Consider POCUS in patients with undifferentiated respiratory distress
- POCUS may identify a ruptured valve causing acute pulmonary oedema and escalate the need for cardiothoracic intervention
- Cardiac ultrasonagraphy should generally reveal some evidence of heart failure for example:
- Systolic heart failure with reduced left ventricular ejection fraction
- Diastolic heart failure may be suggested by left ventricular hypertrophy and a dilated left atrium
- Patients may be hypo/hyper/euvolaemic, therefore, the IVC will not necessarily be distended
Management
- Can remember as LMNOP (backwards) at a junior level but better to approach from n ABC perspective when critically unwell.
Treat Ventilation and Lungs
- Position the patient upright
- Oxygen via NIV (see Settings for congestive heart failure for BiPAP settings)
- HFNP does not improve clinical outcomes significantly
- CPAP or BiPAP2 often starting at 10cm
- Uptitrate CPAP and BiPAP settings accordingly up to 20cm and 24cm/20cm for CPAP and BiPAP respectively
- A trained nurse must remain in attendance at all times as some patients will not tolerate the mask
- Once the patient improves wean the inspired oxygen concentration and PEEP until the patient is comfortable with a normal oxygen mask
- Indicators for intubation:
- Hypercapnia
- pH <7.25 (even in normocapneic patients)
- Severe hypoxia (P/F ratio <190)
- Acute myocardial infarction
- LV ejection fraction below 30%
- Systolic blood pressure <140 mmHg
- If the patient is hypotensive with poor peripheral perfusion, call senior and intensive care (MET Activation Criteria) and treat as for cardiogenic Shock
- Consider therapeutic drainage of a large pleural effusion; doesn’t fix the heart failure but temporally stabilises respiratory function
Optimise MAP
- Nitrates for patients acutely hypertensive:
- Glyceryl trinitrate 10 mcg/minute by IV infusion; increase by doubling the infusion rate every 5 minutes according to clinical response and maintaining systolic blood pressure at >90 mmHg3
- increase cycling of NIBP
- Consider ionotropic support if concomitant cardiogenic shock
- Dobutamine 2.5 to 15mcg/kg/minute by IV infusion
- If the patient is in Atrial Fibrillation with rapid ventricular response, thought to be contributing to poor cardiac output consider:
- Amiodarone 300mg by IV infusion over 30 to 60 minutes
- If still deteriorating consider electrical cardioversion
WARNING
- Beta blockers are contraindicated as they impaire pump function
Optimise Other Factors
- Midazolam or other sedative potentially4
- In patients with evidence of systemic fluid overload, give frusemide (Lasix) 40mg IV or twice the usual daily dose IV (if already on frusemide)
- Many patients with APO have volume redistribution as opposed to fluid overload
- Features which suggest diuresis:
- POCUS shows systemic congestion after controlling SBP
- Hypertension persists despite very high doses of nitroglycerine
- There is a clinical history suggestive of volume overload (e.g. nonadherence with diuretics, missed haemodialysis sessions, increase in weight)
- Physical examination suggests an element of chronic volume overload (e.g. peripheral oedema)
- Other measures to remember:
- Continue oxygen therapy
- Obtain IV access
- Telemetry and other monitoring (blood pressure and pulse oximetry)
- Consider inserting a catheter to monitor urine output
- Also consider:
- Glyceryl trinitrate infusion 10mcg/min; increase by doubling the infusion rate every 5 minutes according to clinical response and maintaining systolic blood pressure at more than 90 mmHg
- Non-invasive ventilation with 100% oxygen: CPAP, starting with 10cm of water pressure
- If difficulty tolerating non-invasive ventilation can consider morphine 1 to 2.5mg intravenously
- If atrial fibrillation with rapid ventricular rate is contributing to poor cardiac output use: amiodarone 300mg by intravenous infusion over 30-60 minutes
- If pulmonary oedema is not responding to the above measures (especially if hypotensive or poorly perfused) consider adding dobutamine 2.5 to 15 mcg/kg/minute by intravenous infusion
Determine precipitating cause for the acute episode
- Six major aetiologies but see list above (Aetiologies)
- Ischaemic heart disease
- Hypertension
- Valvular disease (mitral incompetence, aortic stenosis)
- Cardiomyopathy (dilated, restrictive, hypertrophic) including congenital heart disease
- Pericardial disease
- PE
- The most common precipitating factors include:
- Acute Coronary Syndromes
- Arrhythmia
- Fever, infection
- PE
- Increased sodium or fluid load (parenteral, medicinal, dietary)
- Cardiac depressant drug (e.g. beta-blockers, calcium-channel blockers)
- Sodium-retaining agents (e.g. NSAIDs)
- Renal disease
- Anaemia
- Non-compliance with diet or medication (e.g. dialysis patient)
Sources
- https://coreem.net/core/ape/
- Derranged Physiology
- https://derangedphysiology.com/main/required-reading/cardiovascular-intensive-care/Chapter-230/pulmonary-oedema
- https://derangedphysiology.com/main/required-reading/respiratory-intensive-care/Chapter-993/mechanical-ventilation-pulmonary-oedema
- https://derangedphysiology.com/main/scenarios-blood-gas-interpretation/myocardial-infarction-acute-pulmonary-oedema
- eTG: Acute cardiogenic pulmonary oedema
- https://emcrit.org/ibcc/scape/
- The interpretation of arterial blood gases - Australian Prescriber
- https://www.pocus101.com/lung-ultrasound-made-easy-step-by-step-guide/
Footnotes
-
Apparently RCTs have shown that US is better for diagnosing pulmonary oedema in patients with undifferentiate respiratory distress ↩
-
Doesn’t matter which one, they are both good. This is because it is the PEEP that matters and they both provide PEEP. ↩
-
This is the advice from eTG. Other sources note that above 100mcg/min GTN causes arterial and venous vasodilation which is helpful in the sympathetic crashing acute pulmonary oedema to reduce afterload. I suppose, if unsure about specifically sympathetically driven APO or not conservative may be safer, although as with everything on this website, this is just my own thoughts. ↩
-
Historically in the LMNOP mnemonic, M refers to morphine however retrospective studies have shown that morphine has resulted in increased mortality and ICU admission rate. ↩