Useful information
- With respiratory problems, and pH change in different directions
- With metabolic problems, and pH change in the same direction
Introduction
- Make sure you understand the difference between oxygenation and ventilation
- Have a good understanding of Acute Respiratory Failure
Approach1
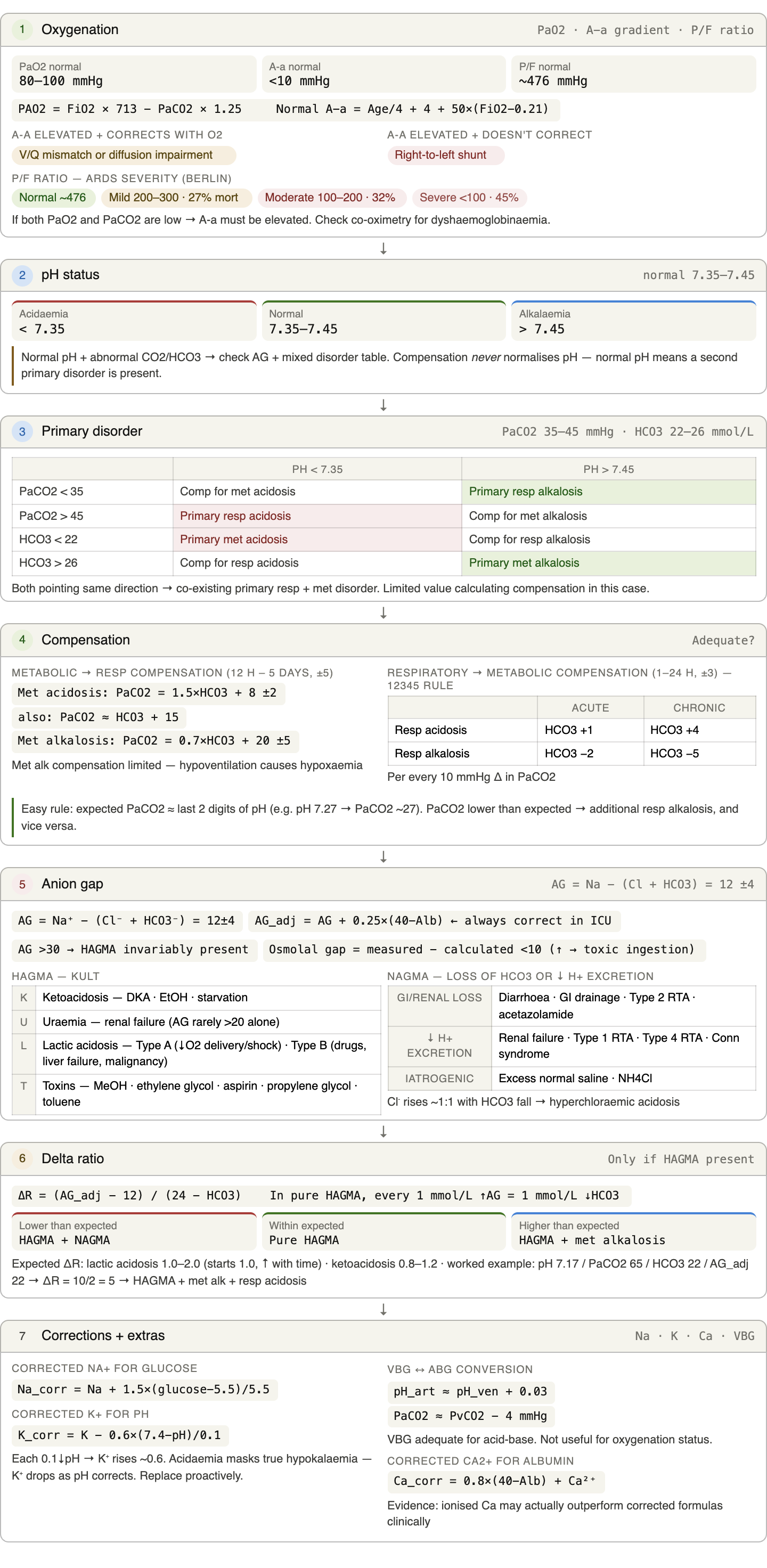
- Determine the adequacy of oxygenation
- Refer to the from the sats probe or as calculated from the ABG or measured from co-oximetry to observe for hypoxaemia
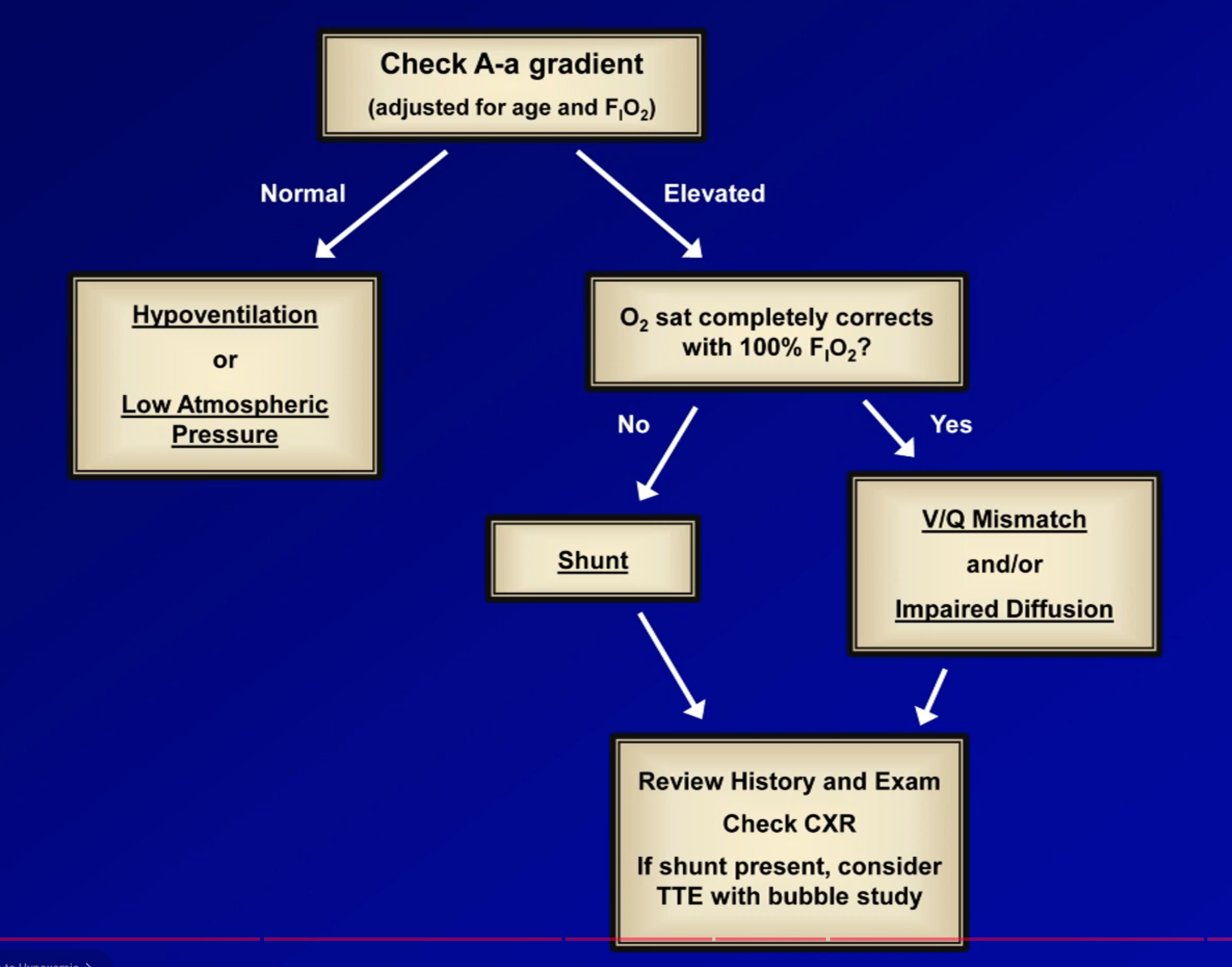
- Calculate the A-a gradient
- Calculate the (see below for calculations and suggested values)
- Calculate using the alveolar gas equation which takes the following values for most patients at sea level
- Calculate the A-a gradient
- Estimate the adjusted normal A-a gradient
- Compare the A-a gradient (<10 mmHg normal)
- If normal, either due to hypoventilation (observe ) or low atmospheric pressure
- If elevated, observe for correction of sats with 100%
- If corrects, suggestive of V/Q mismatch and/or diffusion impairment
- If doesn’t correct, suggestive of right to left shunt
- Correlate with history, exam and chest X-ray
- Calculate the ratio in ARDS patients where:
- 300-200 ⇒ mild ARDS with 27% mortality
- 200-100 ⇒ moderate ARDS with 32% mortality
- <100 ⇒ severe ARDS with 45% mortality
- Note that it can be corrected for PEEP with the following formula:
- Note only use when is normal and a shunt is not expected
- Check for dyshaemoglobinaemia from the co-oximetry
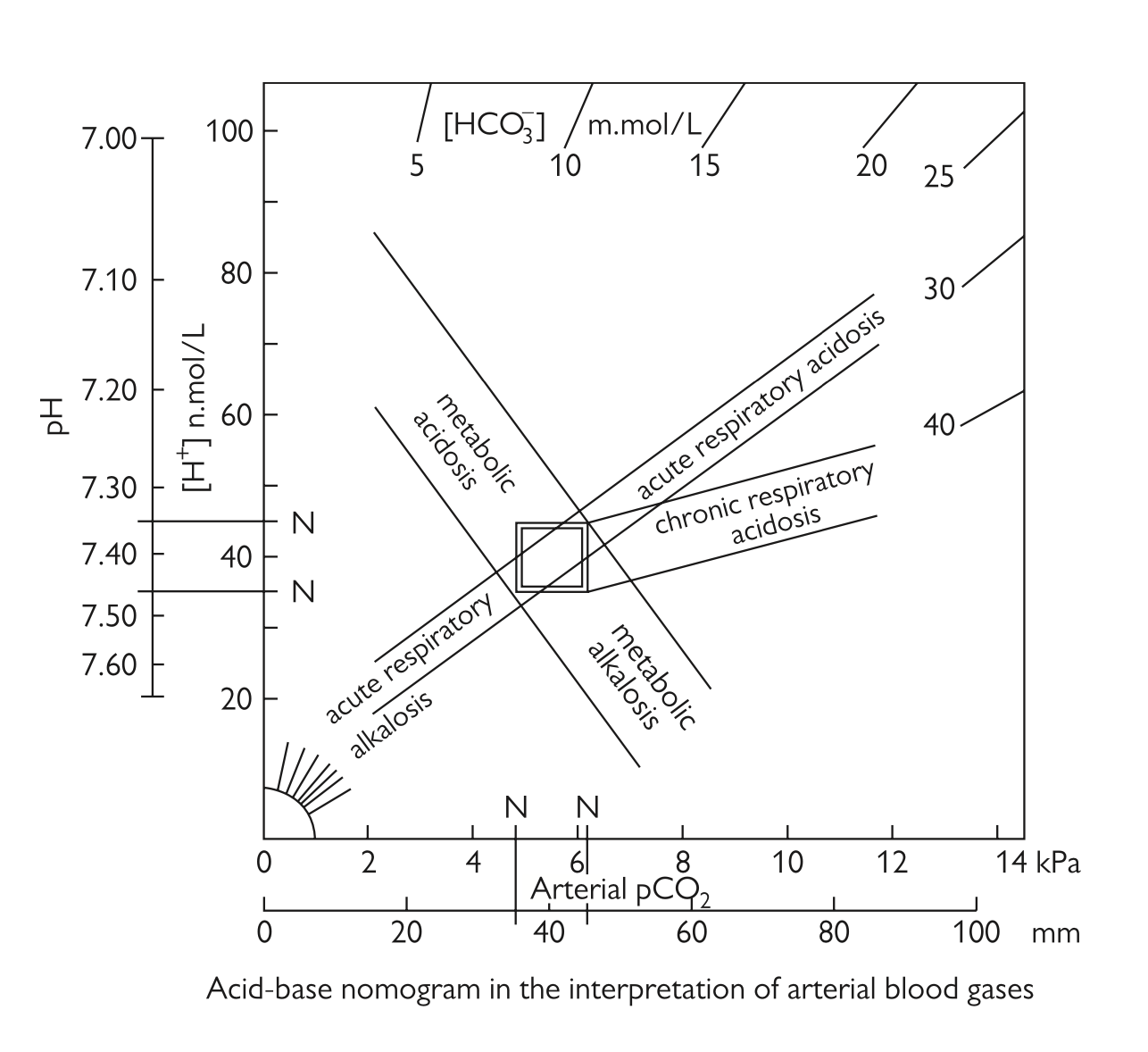
- Determine pH status (7.35-7.45)
- pH <7.35: acidaemia: increase in the serum hydrogen ion concentration, lowers the pH
- pH >7.45: alkalaemia: decreases the hydrogen ion concentration, raises the pH
- If pH is normal and or is abnormal check the anion gap and the table on mixed disorders below
- Determine the primary disorder
- : Determine the respiratory component (35-45 mmHg)
- <35 ⇒ ↑ respiratory rate ⇒ alkalotic change
- If pH >7.45: primary respiratory alkalosis (especially if normal)
- If pH <7.35 and ↓ : respiratory compensation for metabolic acidosis
- >45 ⇒ ↓ respiratory rate ⇒ acidotic change
- If pH <7.35 and normal: hypoventilation
- If pH >7.45 and ↑ : respiratory compensation for metabolic alkalosis
- <35 ⇒ ↑ respiratory rate ⇒ alkalotic change
- : Determine the metabolic component (22-26 mmol/L)
- <22 ⇒ acidotic change
- If pH <7.35: primary metabolic acidosis
- If pH >7.45: metabolic compensation for respiratory alkalosis
- >26 ⇒ alkalotic change
- If pH >7.45: primary metabolic alkalaosis
- If pH <7.35: metabolic compensation for respiratory acidosis
- <22 ⇒ acidotic change
- If both and are causing the same disturbance (i.e. both acidotic or alkalotic changes) there is co-existing primary respiratory and metabolic disorders
- There is limited utility in calculating expected compensation
- : Determine the respiratory component (35-45 mmHg)
- Is the compensation adequate
- If the pH is normal, there is another primary acid/base disorder as compensation will never bring the pH back to adequacy (e.g. if there is respiratory acidosis and the pH is normal, there must also be a primary metabolic alkalosis)
- Metabolic compensation takes 12 hours - 5 days to take peak effect (generally for metabolic compensation formulas)
- Metabolic acidosis: (Winter’s formula) or
- Metabolic alkalosis: or
- Compensation for a metabolic alkalosis is relatively poor because hypoventilation as a compensation mechanism results in hypoxaemia
- Easier rule for compensation in metabolic disorders: is approximately the same as the first two digits of the pH after the decimal point
- e.g ABG: pH 7.27 / 25 / 11 ⇒ decimals of pH is 27 and is 25 so approximately equal, hence physiologic compensation is adequate
- If the is lower than expected then there is additional respiratory alkalosis and vice versa
- Respiratory compensation takes 1 - 24 hours to take peak effect (generally for respiratory compensation formulas)
- Respiratory acidosis
- Acute: or
- Chronic: or
- Respiratory alkalosis
- Acute: or
- Chronic: or
- Respiratory acidosis
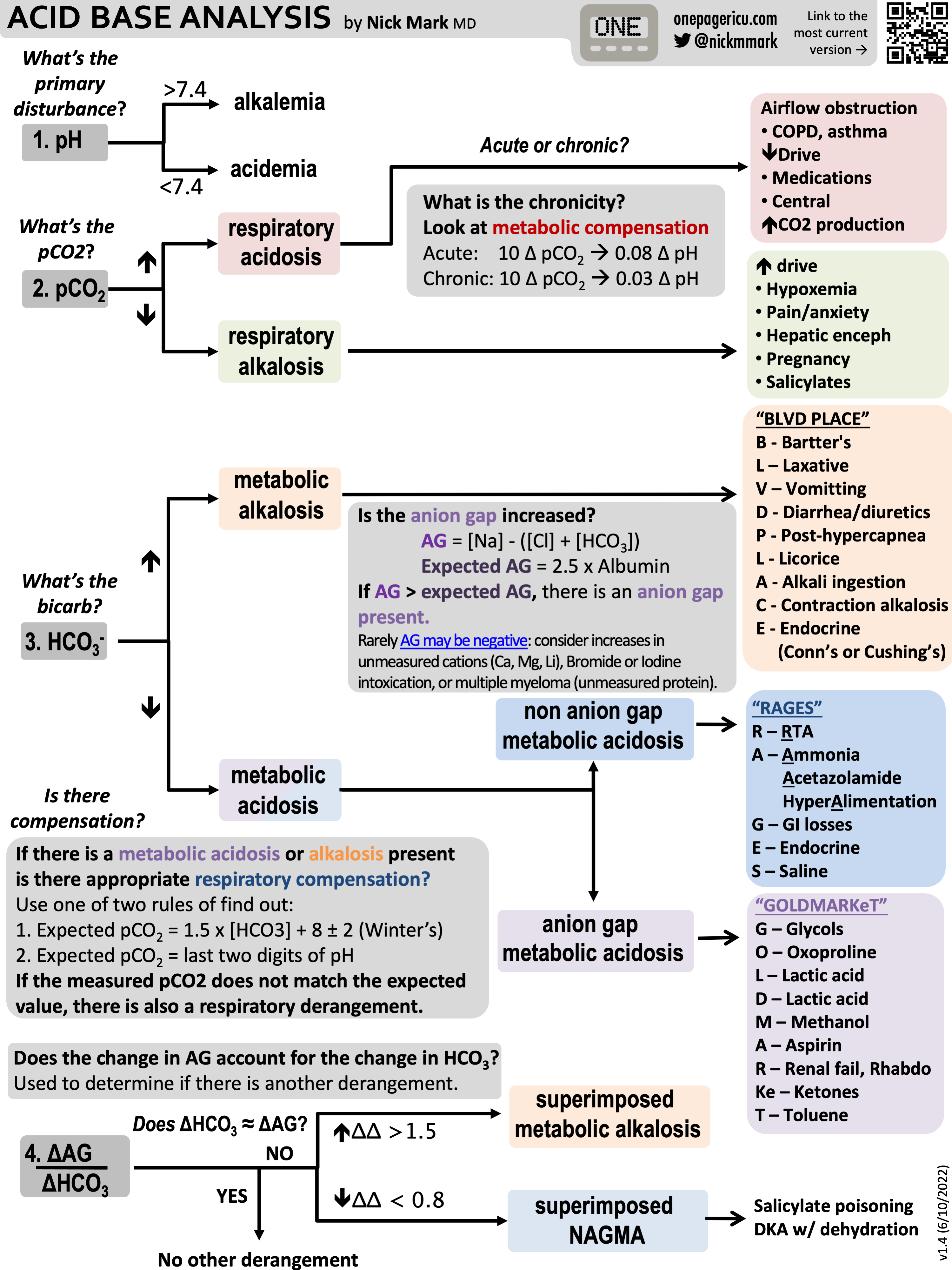
- Calculate the anion gap
- High anion gap acidosis ⇒ addition of anions (conjugate bases of acids added to serum e.g. ketones or lactate)
- Severe renal failure can lead HAGMA to excrete phosphate and sulphate in the urine resulting in their accumulation
- Lactic acidosis, ketoacidosis (DKA, ETOH, starvation), methanol ingestion, ethylene glycol ingestion, toluene inhalation (glue sniffing)
- Normal anion gap acidosis ⇒ decreased renal excretion or loss of in urine or GI tract
- Loss of : diarrhoea/GI drainage, Type 2 Renal tubular acidosis, Acetazolamide
- Decreased excretion: renal failure, Type 1 Renal tubular acidosis, Type 4 Renal tubular acidosis/Conn syndrome
- Results in decrease in with an increase in a roughly 1:1 ratio (hence NAGMA sometimes called hyperchloraemic acidosis)
- Potassium is not included because it tends to influence the result little2 however if one insisted, the normal value would be around 16
- Anion gap unrelated to metabolic acidosis:
- High anion gap can be due to metabolic alkalosis, hyperphosphataemia
- Low anion gap can be due to hypoalbuminaemia, ↑ , ↑ , ↑ , Severe lithium toxicity, bromide ingestion (some machines mistake bromine for chlorine; Myaesthenia gravis medications can contain bromine)
- Adjust anion gap for hypoalbuminaemia3:
- High anion gap acidosis ⇒ addition of anions (conjugate bases of acids added to serum e.g. ketones or lactate)
- Delta ratio which should be calculated whenever the anion gap is elevated
- During a HAGMA there should be a 1:1 gap between the ↑ anion gap and ↓
- Check delta ratio in the presence of a high anion gap metabolic acidosis to determine if it is a pure HAGMA or if there is co-existent normal anion gap metabolic acidosis or metabolic alkalosis
- The expected delta radio differs by aetiology of HAGMA:
- Lactic acidosis: 1.6 (1.0 - 2.0) with it initially starting at 1.0 and increasing with time
- Ketoacidosis: 1.0 (0.8 - 1.2) unless ↓ GFR in which case the ratio may be higher
- Kidney disease: variable depending on the extent of tubular damage relative to ↓ GFR
- Methanol, ethylene glycol ingestion: Unknown, probably 1.0 - 2.0
- Based on the above, the interpretation is made such that if the measured delta ratio is:
- Lower than the expected range, there is HAGMA and NAGMA
- Within expected range, there is HAGMA only (although there could be respiratory disorder)
- Higher than the expected range, there is a HAGMA and metabolic alkalosis
- A negative delta ratio is possible in the following contexts:
- High anion gap metabolic acidosis and metabolic alkalosis (can result in a normal or high pH)
- Mild high anion gap metabolic acidosis and severe chronic respiratory acidosis (usually results in a very low pH)
- An alternative easier calculation is the delta gap, where the interpretation is as follows
- Less than -6 ⇒ mixed high and normal anion gap acidosis
- -6 to 6 ⇒ only high anion gap acidosis exists
- More than 6 ⇒ mixed high anion gap acidosis and metabolic alkalosis
- Apply corrections to certain measured values
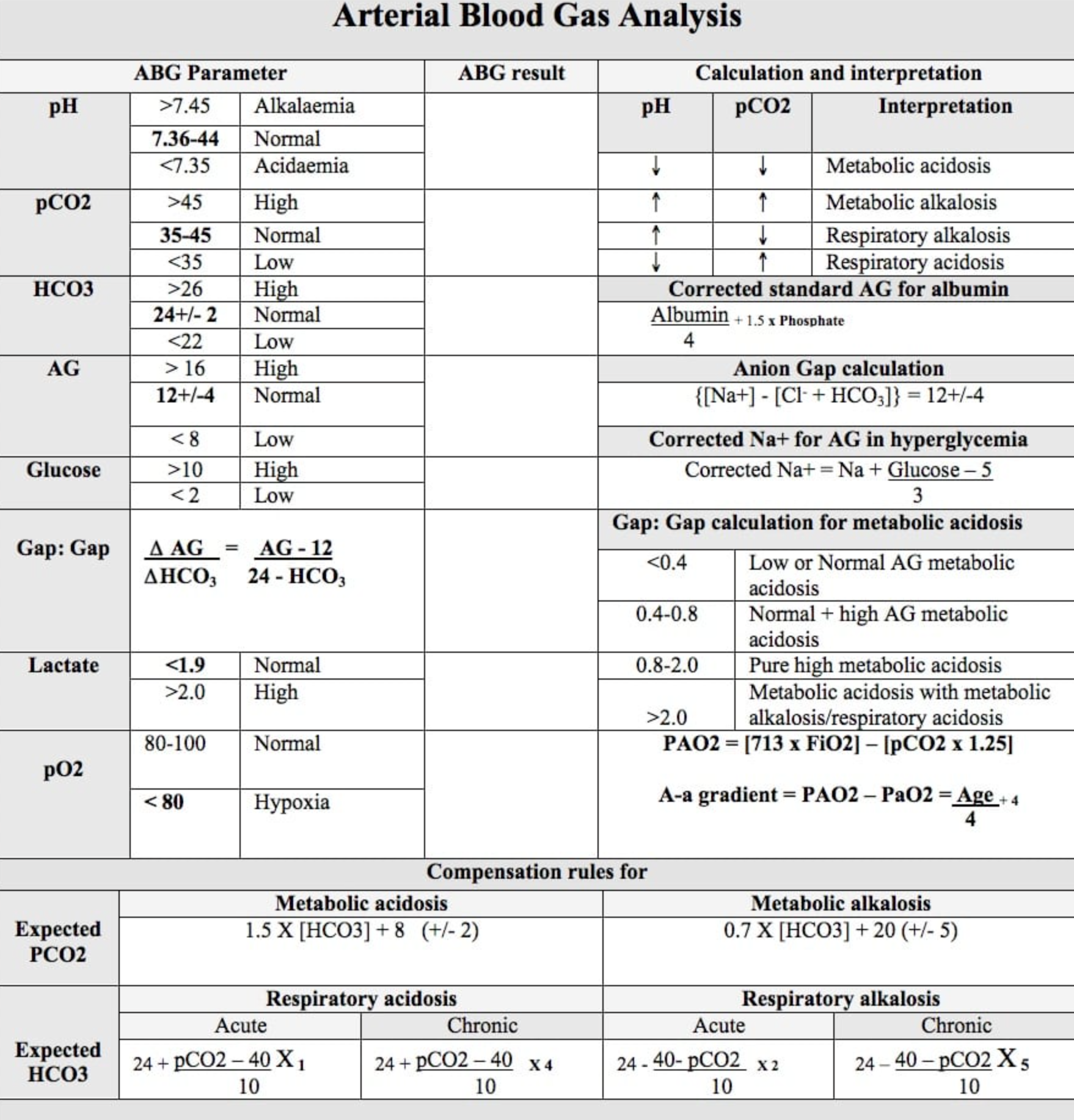
- As an extra step you can determine the consistency of the values from the machine and confirm that the derrived is correct using:
Additional Details
Differentials
High Anion Gap Metabolic Acidosis
- Mnemonic: KULT
- Ketoacidosis
- Diabetes (see: Diabetic ketoacidosis)
- Starvation
- Chronic alcoholism
- Uraemia
- Renal failure
- Occurs for 2 reasons:
- Failure to excrete ions in the kidney due to ↓ excretion of and ↓ excretion of titratable acids (e.g. )
- Accumulation of unmeasured anions (phosphate, sulphate, urate and hippurate)
- The anion gap from renal failure rarely exceeds 20 and often co-exists with normal anion gap acidosis
- Occurs for 2 reasons:
- Renal failure
- Lactic acidosis
- Increased production of lactate:
- Problem with delivery (e.g. shock, bowel infarction, hypoxaemia)
- Problem with utilisation (e.g. genetic enzymatic defects, mitochondrial toxins, thiamine deficiency)
- Primary production from tumours (e.g. lymphone, leukaemia and multiple myeloma)
- Increased motor activity (e.g. seizures, vigorous exercise)
- Decreased clearance (e.g. liver failure or renal failure although noting that renal failure alone is unlikely to cause lactic acidosis)
- GI absorption of D-Lactate (usually in patients who have undergone either jejunoileal bypass or extensive resection of small bowel)
- Typically presents as elevated anion gap acidosis that appears after eating and resolves with fasting (clinical appearance of altered mental status)
- Measured lactate levels are normal unless specifically testing for D-lactate
- Medications (e.g. paracetamol, anti-retrovirals, beta agonists, 5-flurouracil, halothane, iron, isoniazid, linezolid, nitroprusside, propofol, salicylates, sorbitol, sulfasalazine, valproic acid)
- There remains disagreement about metformin causing lactic acidosis
- Toxins (e.g. carbon monoxide, cocaine, cyanide, diethyl ether, ethanol, toxic acidosis)

- Increased production of lactate:
- Toxins
- Methanol
- Found in windshield wiper fluid, antifreeze, paint remover
- Presents with vision loss, photophobia, abdominal pain, confusion and lethargy
- Ethylene glycol
- Found in antifreeze and liquid coolant
- Presents with
- Confusion at <12 hours
- Heart failure, myocarditis and pulmonary oedema at 24 hours
- Acute kidney injury at 24-72 hours
- Also occurs lactic acidosis as glycolate impairs cellular respiration causing lactic acidosis
- Can be diagnosed via calcium oxalate crystals in the urine (can be quicker to obtain than ethylene glycol level)
- Ethanol
- Propylene glycol
- Used as a solvent for lorazepam, phenobarbital, diazepam, phenytoin
- Presents with renal failure and unexplained lactic acidosis in a patient who has been on IV infusion of lorazepam for several days
- Recommended limit of propylene glycol is 69g/day (equivalent to 7mg/hr of lorazepam infusion)
- Toluene
- Found in glues/adhesives and paint thinner
- Acutely presents with euphoria, loss of inhibition, amnesia, slurred speech and ataxia
- Chronically it can cause cerebellar dysfunction, dementia, rental tubular acidosis, hypokalaemia and renal failure
- Salicylic acid (aspirin)
- 5-oxoproline
- Associated with chronic paracetamol use with additional risk factors including advanced age, malnutrition, chronic illness and alcoholism
- Symptoms are non-specific
- Methanol
- Ketoacidosis
- Approach
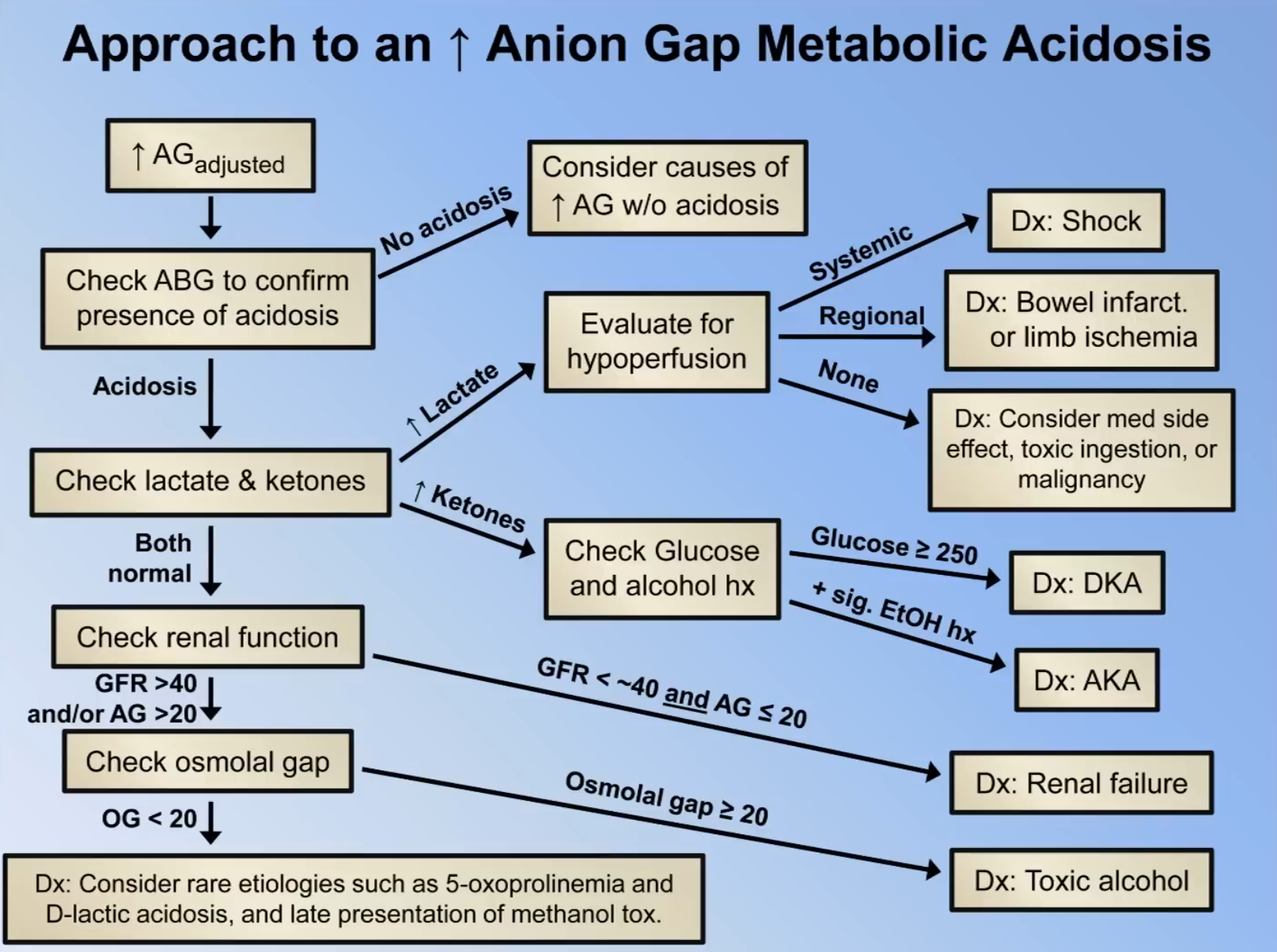
- Check anion gap and adjust for hypoalbuminaemia
- Check ABG to ensure acidosis is present (i.e. low bicarb); if no acidosis review causes of ↑ AG w/o acidosis (e.g. hyperphosphataemia, anionic paraprotein)
- Check lactate and ketones
- If ↑ lactate, evaluate for hypoperfusion
- If systemic hypoperfusion it suggests Shock
- If regional hypoperfusion, probably related to that (e.g. bowel infarct)
- If nil hypoperfusion consider medical side effect, toxic ingestion or lactate producing malignancy
- Any altered mental status or Hx of toxic ingestion check: paracetamol level, osmolality + osmololal gap, urine toxicology screen, aspirin level
- Causes of increased osmolal gap include ethanol, methanol, ethylene glycol and isopropanol ingestion
- If ↑ ketones, check glucose and alcohol Hx
- Glucose ≥ 13.9 suggests Diabetic ketoacidosis
- If Hx of ETOH suggests alcoholic ketoacidosis
- If ↑ lactate, evaluate for hypoperfusion
- Check renal function (if above normal)
- If GFR < 40 and AG ≤ 20 ⇒ renal failure alone is likely cause, otherwise check osmolal gap
- Check rare aetiologies such as D-lactic acidosis and late presentation of methanol toxicity
- General indications for calculating the osmolal gap are:
- Suspected posining with unknown toxin
- Elevated anion gap in the presence of normal lactate, ketones and renal function
- Unexmplained altered mental status (particularly in an alcoholic or child)
- Periodic monitoring for patients on high doses of IV lorazepam
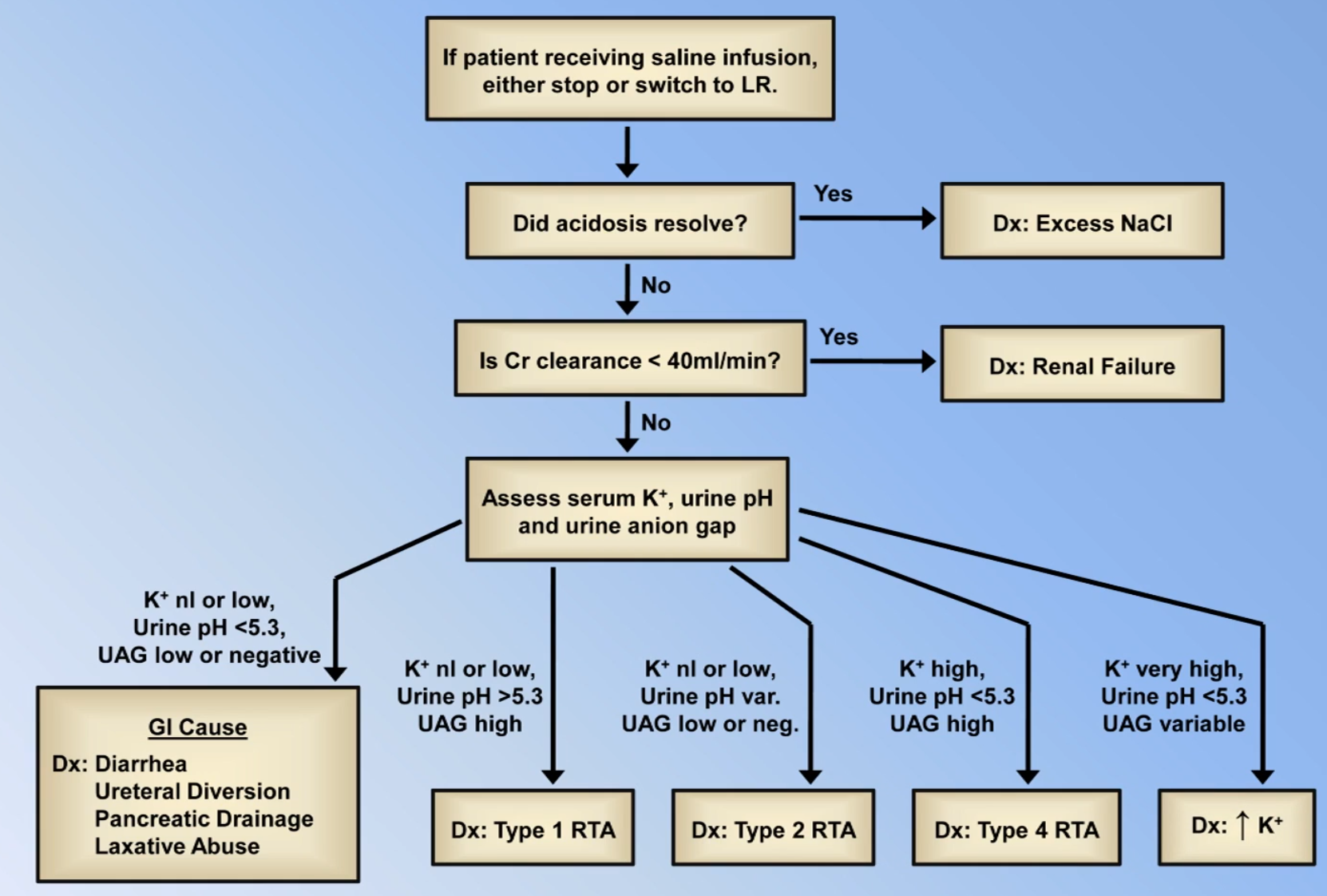
Normal Anion Gap Metabolic Acidosis
| Primary Issue | GI Problem | Renal Problem |
|---|---|---|
| Gain of or failure to excrete | Hyperalimentation (e.g. too fast TPN) | Types 1 and 4 RTA Renal failure Hyperkalaemia |
| Loss of | Diarrhoea External pancreatic drainage Ureteral diversion Oral Cholestyramine | Type 2 RTA |
- Other causes: Infusion of normal saline or
Metabolic Alkalosis
| Primary Issue | GI Problem | Renal Problem |
|---|---|---|
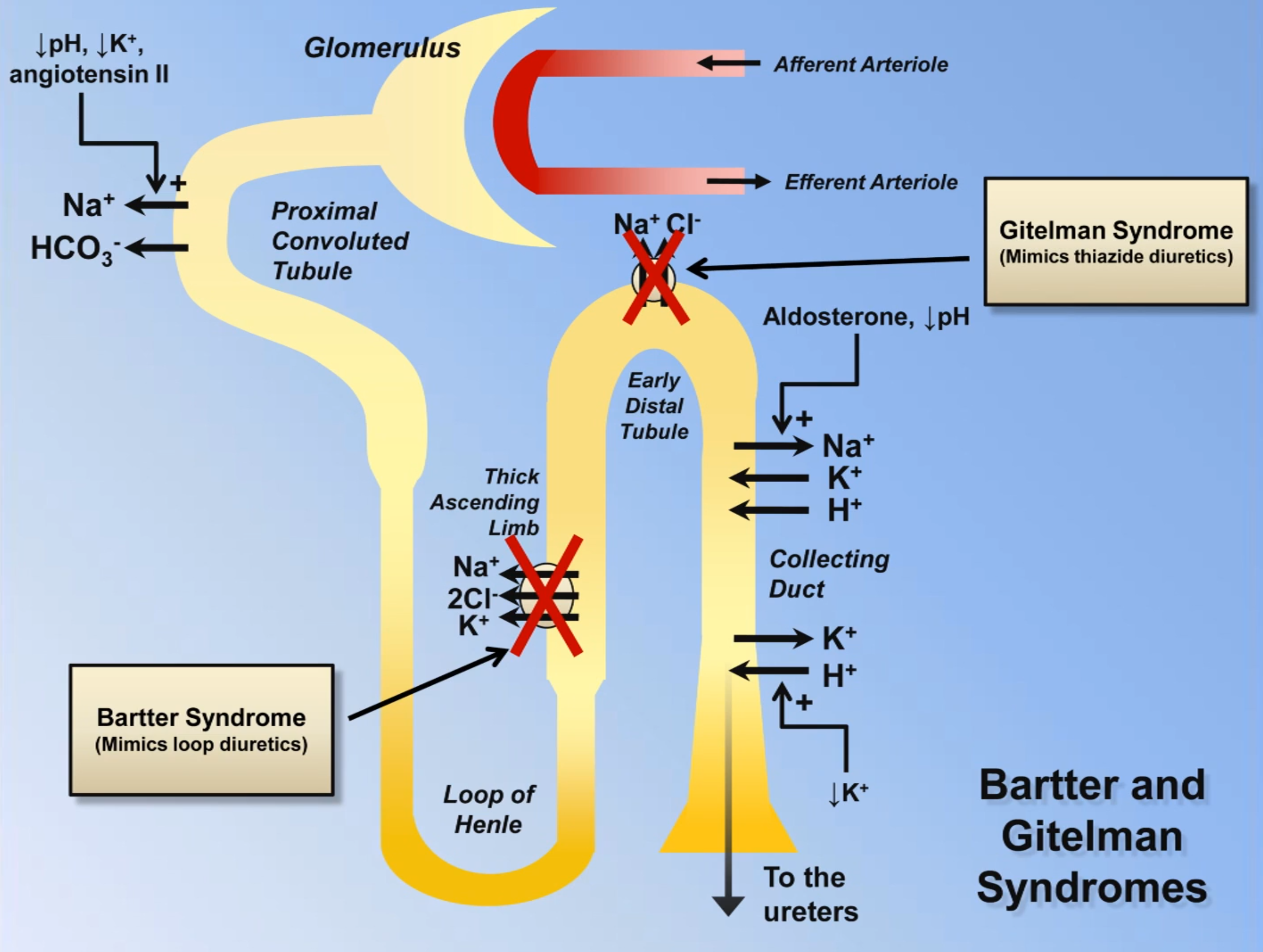
| Loss of | Vomiting NG suction Congenital chloride diarrhoea | Loop/Thiazide Diuretics Mineralocorticoid Excess Contraction Alkalosis Bartter/Gitelman Syndromes |
| Gain of | Milk-alkali syndrome Ingestion of | Contraction Alkalosis |
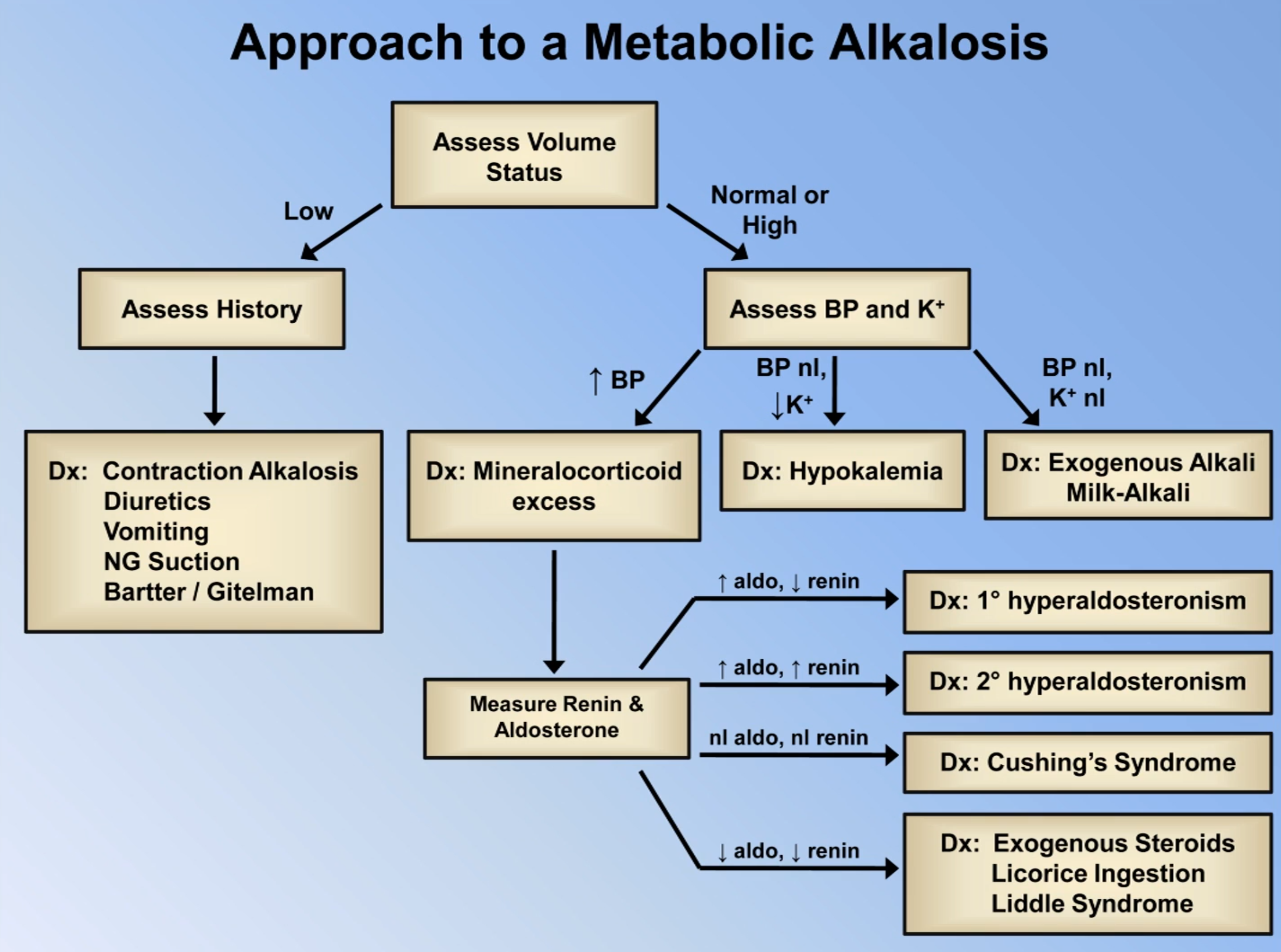
- Contraction alkalosis
- When intravascular volume depletion occurs → low renal volume perfusion → ↑ aldosterone and angiotensin II → ↑ reabsorption ↑ reabsorption, ↑ excretion, ↑ excretion → metabolic alkalosis
- Diuretics
- Can result in contraction alkalosis
- By inhibiting sodium reabsorption proximally in the nephron, more sodium reaches the collecting duct
- This results in increase in sodium reabsorption and excretion (see Diuretics)
- Vomiting/NG suction
- Volume depletion causing a secondary contraction alkalosis
- Direct loss of in gastric fluids
- Mineralocorticoid excess (suggested by hypertension, hypokaelamia, metabolic alkalosis)
- Hyperaldosternism (e.g. from primary hyperaldoserism as in Conn’s syndrome or from secondary hyperaldosterism due to elevated from renin as in renal artery stenosis)
- Cushing’s syndrome (e.g. CRH producing tumour as in bronchial carcinoid tumours, elevated ACTH as in pituitary adenoma termed Cushing’s disease or elevated cortisol in an adrenal adenoma or exogenous use)
- Hypokalaemia
- Leads to a shift of from intracellular to extracellular space in exchange for a shift of from extracellular to intracellular space
- Within the nephron, hypokalaemia stimulates reabsorption in the PCT and excretion in the CD
- Milk-Alkali syndrome (characterised by hypercalcaemia, metabolic alkalosis and renal insufficiency)
- Occurs when large amount of calcium and absorbable alkali consumed
- Common in women taking calcium supplements
- Bartter & Gitelman sydrome (shared features: metabolic alkalosis, hypokalaemia, high renin and aldosterone and lack of hypertension)
Respiratory Acidosis
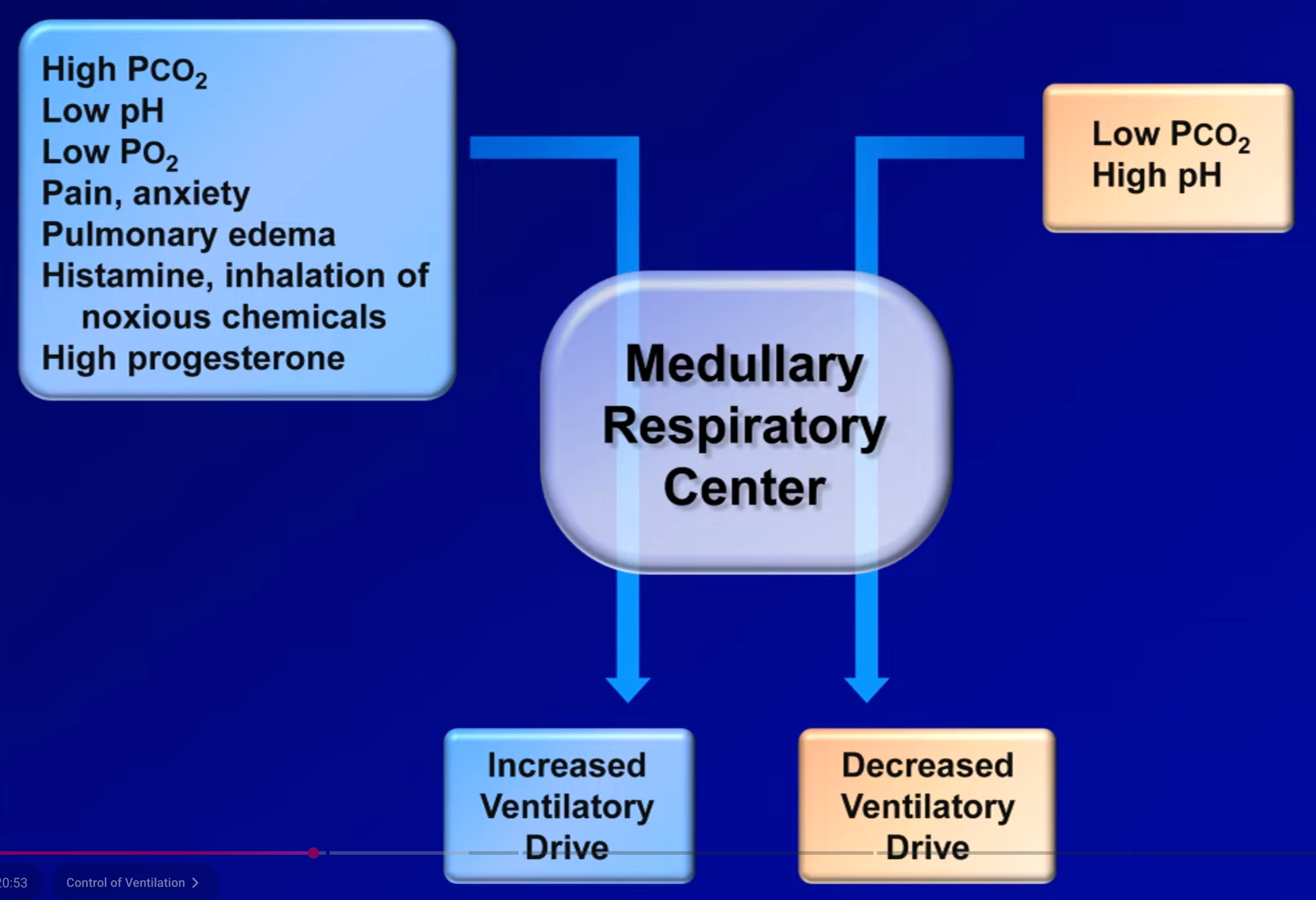
- Alveolar ventilation equation:
- Where:
- is the rate of production
- is the total pressure of inspired air
- is alveolar ventilation
- is the tidal volume
- is the dead space
- It therefore follows that the causes of ↑ can be due to low respiratory rate, low tidal volume, high dead space or high rate of production

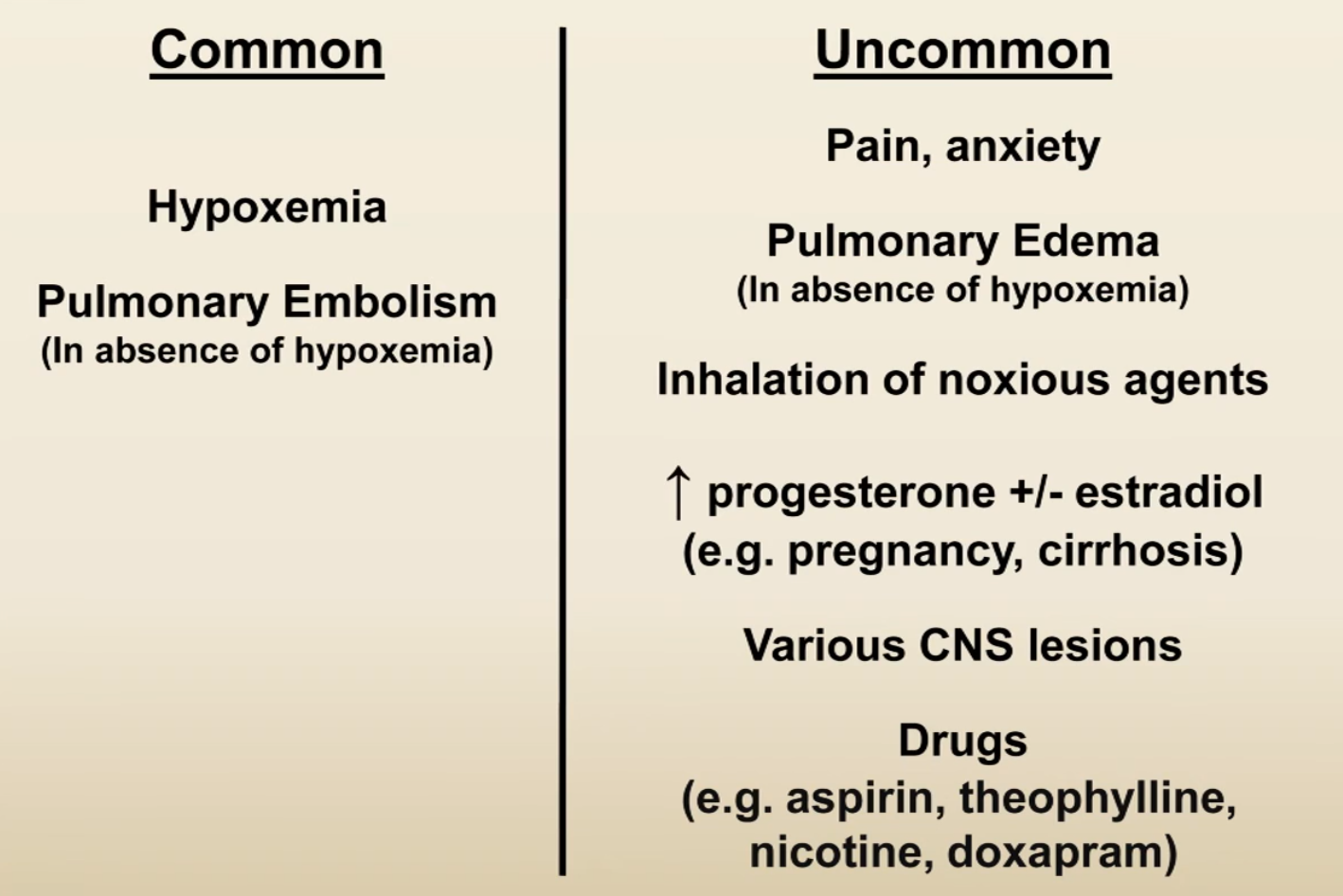
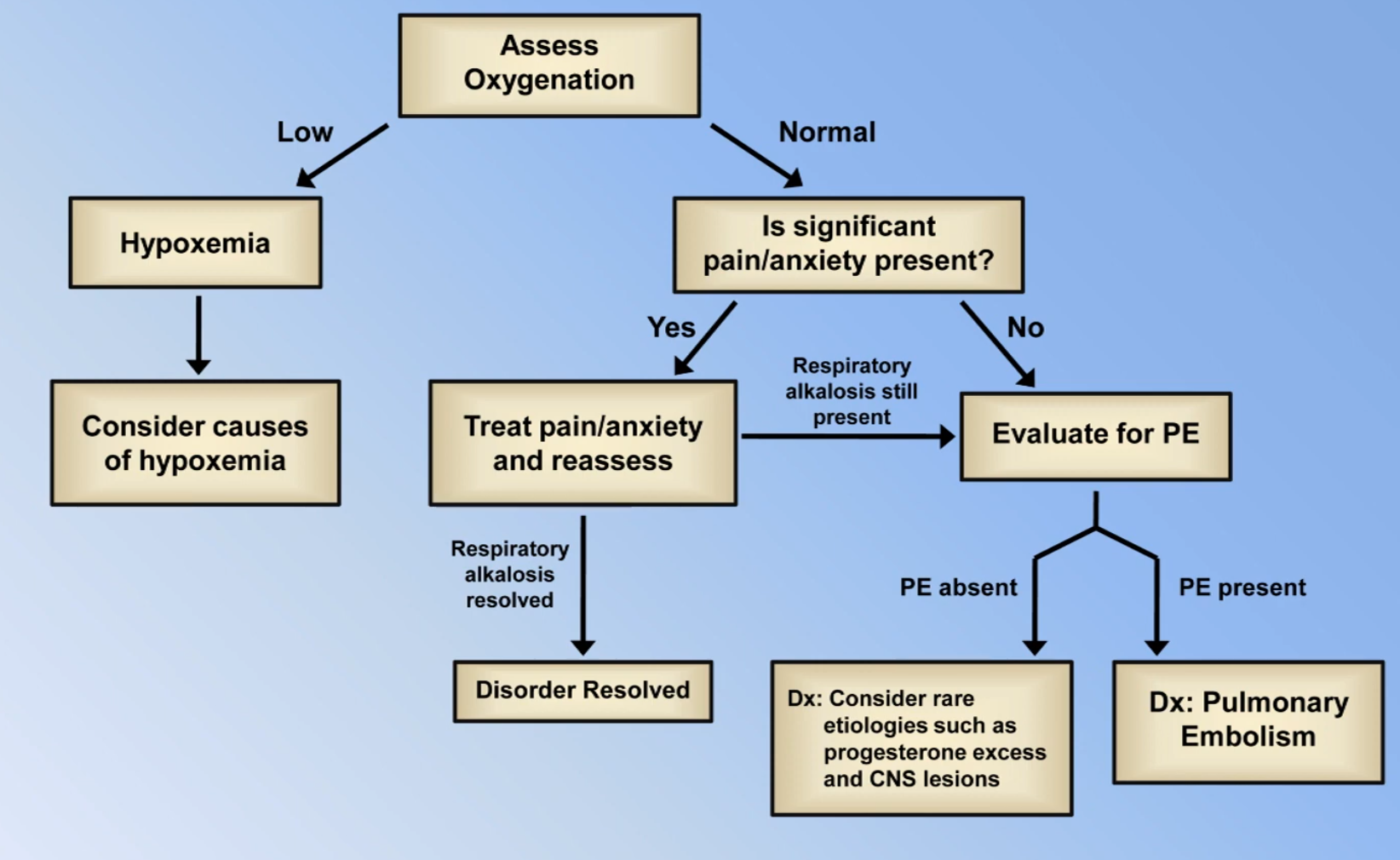
Respiratory Alkalosis
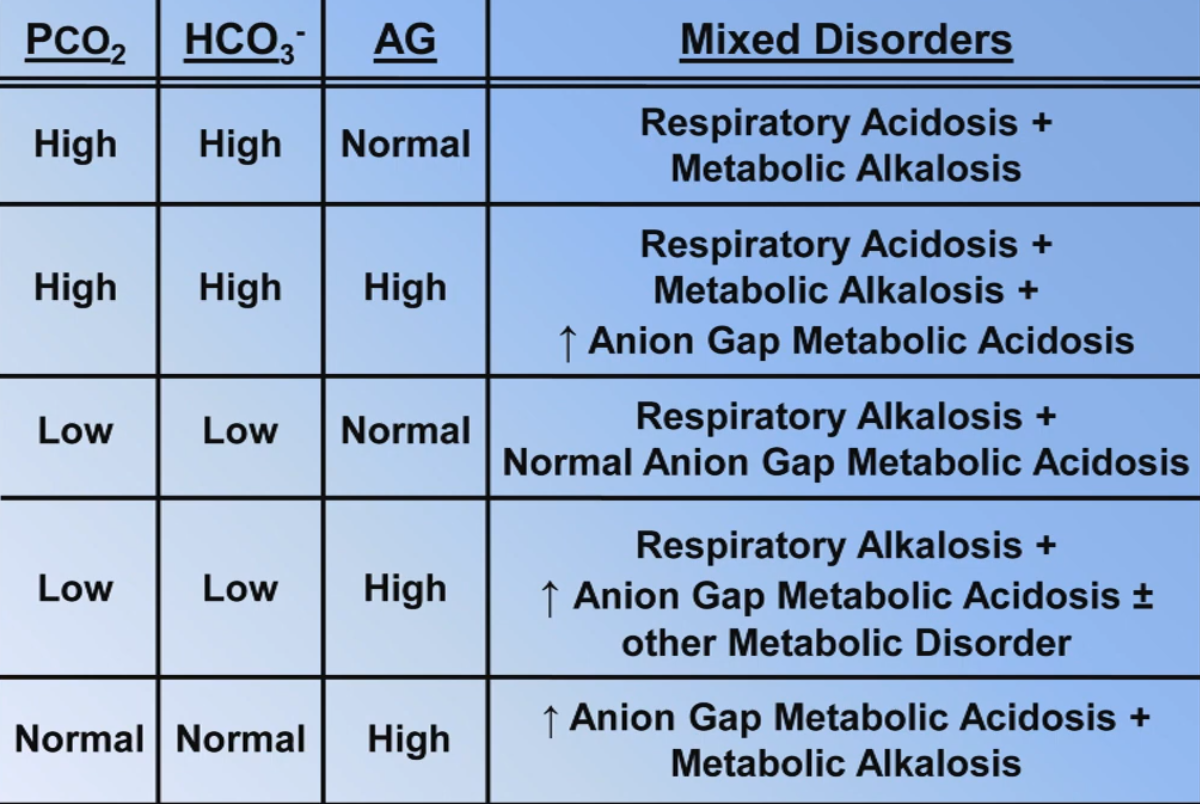
Patterns of Mixed Disorders with Normal pH
Drug Overdose Changes
- Respiratory alkalosis in the setting of drug overdose can be due to:
- Aspiration on vomit causing hypoxia induced hyperventilation
- Aspirin ingestion causing hyperventilation
- Most drug overdose causes respiratory acidosis
A-a Gradient and Oxygenation

- Hypoxia = pathophysiological state of inadequate oxygenation for aerobic metabolism; aetiologies include hypoxaemia, anaemia, dyshaemoglobinaemia, histotoxic hypoxia (e.g. cyanide poisoning)
- Hypoxaemia = ↓ concentration of oxygen in arterial blood
- is the amount of oxygen dissolved in the blood and is measured directly from the ABG (normal >85 mmHg for young adult, child and >75 mmHg for elderly)
- sat commonly refers to the oxygen bound to haemoglobin:
- is the amount of oxygen bound to haemoglobin as measured from pulse oximetry
- is the amount of oxygen bound to haemoglobin as calculated from the ABG or measured by co-oximetry
- is the partial pressure of oxygen in the alveolar gas
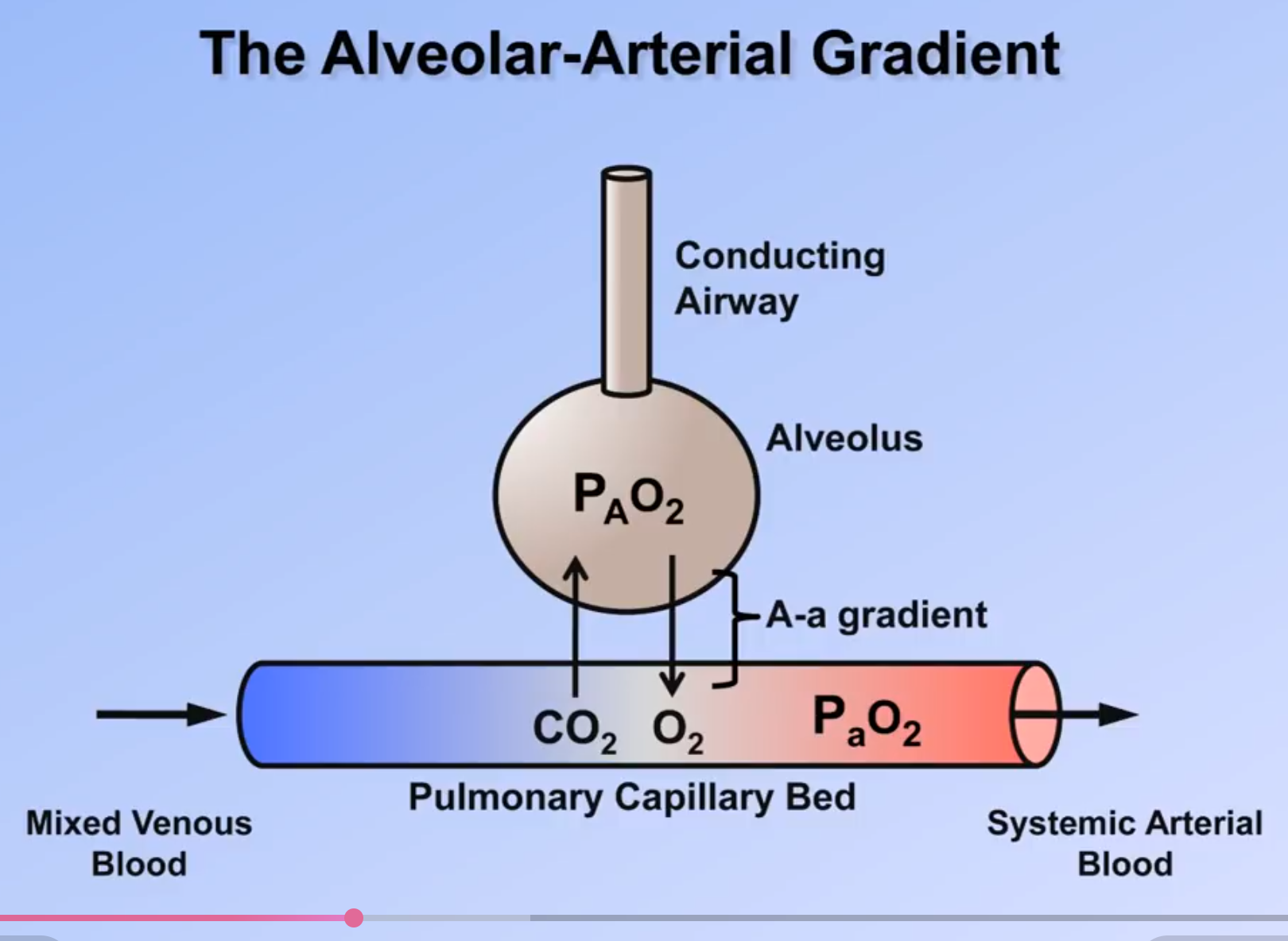
- At sea level (when not on oxygen), , and for most patients 4so the above equation simplifies to:
| Oxygen Supply | Approximate Maximum |
|---|---|
| Room air | 0.21 |
| Nasal cannula | 0.50 |
| Venturi mask | 0.50 |
| Simple face mask | 0.60 |
| Non-rebreather | 0.80-0.90 |
| Mechanical ventilation | >0.90 |
- Variation of with flow rate and respiratory rate for nasal prongs:
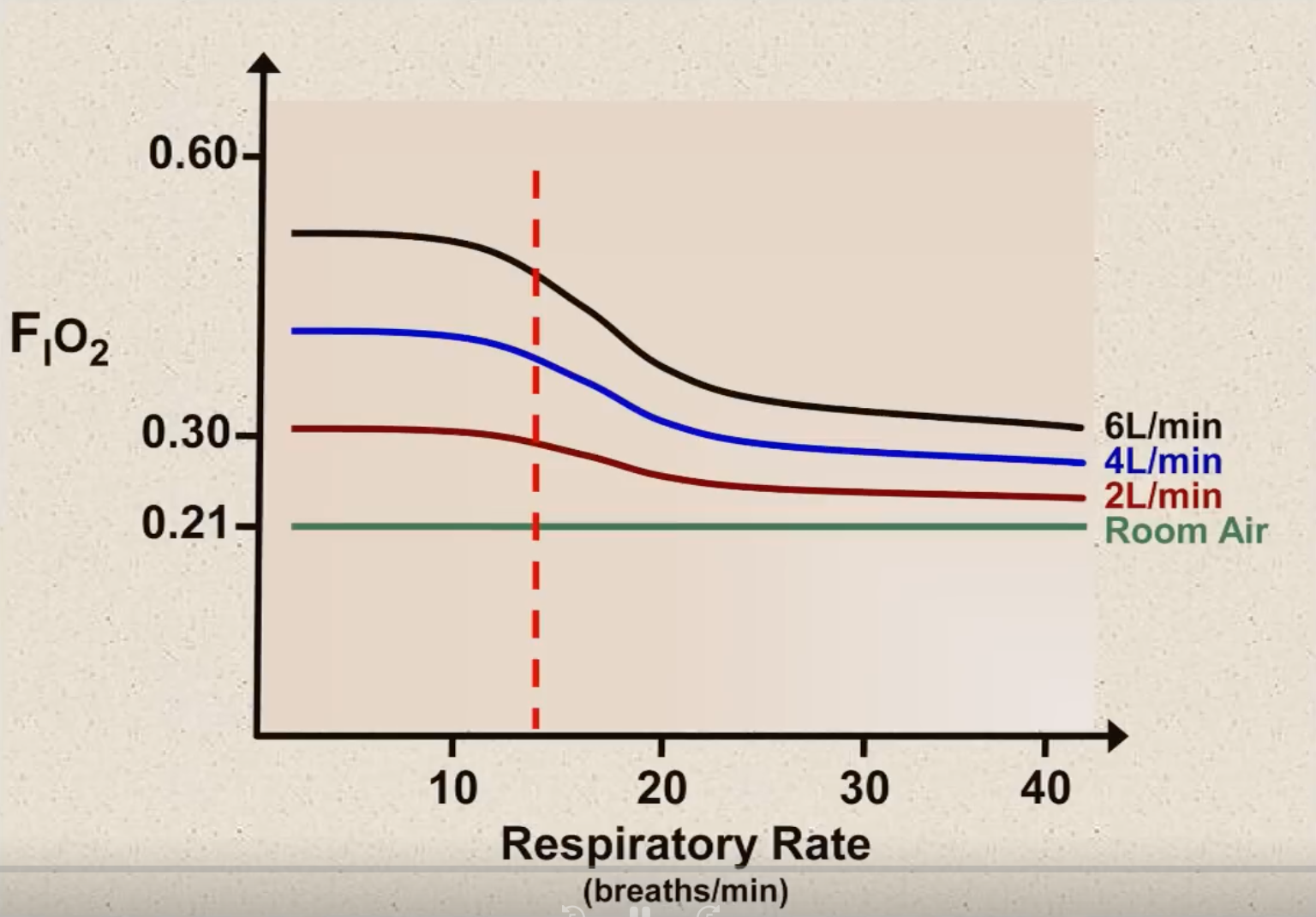
- Can be estimated with the following formula (use 0.03 for patients with increased respiratory rate and 0.05 for patients with normal respiratory rate):
- Variation of with respiratory rate
- High flow systems are able to meet the patient’s full inspiratory flow requirement (i.e. is independent of respiratory rate) (e.g. venturi mask, HFNP, mechanical ventilation)
- Low flow systems are not able to meet the full inspiratory flow requirement (i.e. varies with respiratory rate)
- The normal A-a gradient increases with age5 and can be calculated by the following equation when there is no supplemental oxygen:
- Due to physiological shunting caused by supplemental oxygen, the adjust A-a gradient can be approximated by:
NOTE
If a patient has both a low and a low , then the A-a gradient must be elevated
- If the A-a gradient is normal then the cause of the hypoxaemia must be either:
- Hypoventilation (i.e. ↑ )
- Low (e.g. extreme altitudes)
- If the A-a gradient is elevated then the cause of the hypoxaemia must be either:
- V/Q mismatch
- Shunt (i.e. there exists perfused alveoli with zero ventilation e.g. cardiac shunts, lobar pneumonia ARDS)
- Importantly when is increased, there is an increase in the A-a gradient as can not be further increased
- Impaired diffusion
Simple Example
Question: A 56 year old man with a history of CAD, HTN, and 60 pack years of smoking, presents to the ER with a productive cough and dyspnea x 3 days. On exam, his RR=28 and 02 sat = 81% on room air. His breaths are shallow and with pursed lips. ABG: pH: 7.31 / 60 / 57 on room air Answer:
- Check A-a gradient
- Estimate normal A-a gradient
- Normal A-a gradient with high indicates a COPD exacerbation
Harder Example
Question: An 80 y/o man with COPD, presents to the ER with progressive dyspnea and cough for 3 days. He appears acutely uncomfortable, and his sitting up, leaning forward with his hands on his knees. Vitals: T=36.9°C, HR=104, BP=135/80, RR=30, 02 saturation=95% on Venturi mask set at 30%. ABG: pH: 7.28 / 80 / 80 on 30% Answer:
- Check A-a gradient
- Estimate adjusted normal A-a gradient
- Pretty much normal A-a gradient with high indicates an acute COPD exacerbation
Harder Example
Question: A 64 y/o man with a history of CAD, presents to the ER with sudden onset of dyspnea 1 hour ago. He is in moderate respiratory distress, with rapid, shallow breaths. Current vitals: T=36.2 °C HR=115, BP=95/40, RR=30, O2 saturation=100% on 6L via nasal prongs. ABG: pH 7.54 / 28 / 143 on 6L via NP Answer:
- Check the A-a gradient
- Estimate adjusted normal A-a gradient
- Abrupt changes in A-a gradient without symptoms suggestive of hypertensive emergency or mucous plugging can be explained by pulmonary embolism or acute coronary syndrome with secondary pulmonary oedema
Harder Example
- Increase in A-a gradient with increase suggests right to left shunting
- Patient appears to be due to intrapulmonary right to left shunting in a patient with septic shock (most likely severe sepsis and acute lung injury/ARDS secondary to endocarditis)
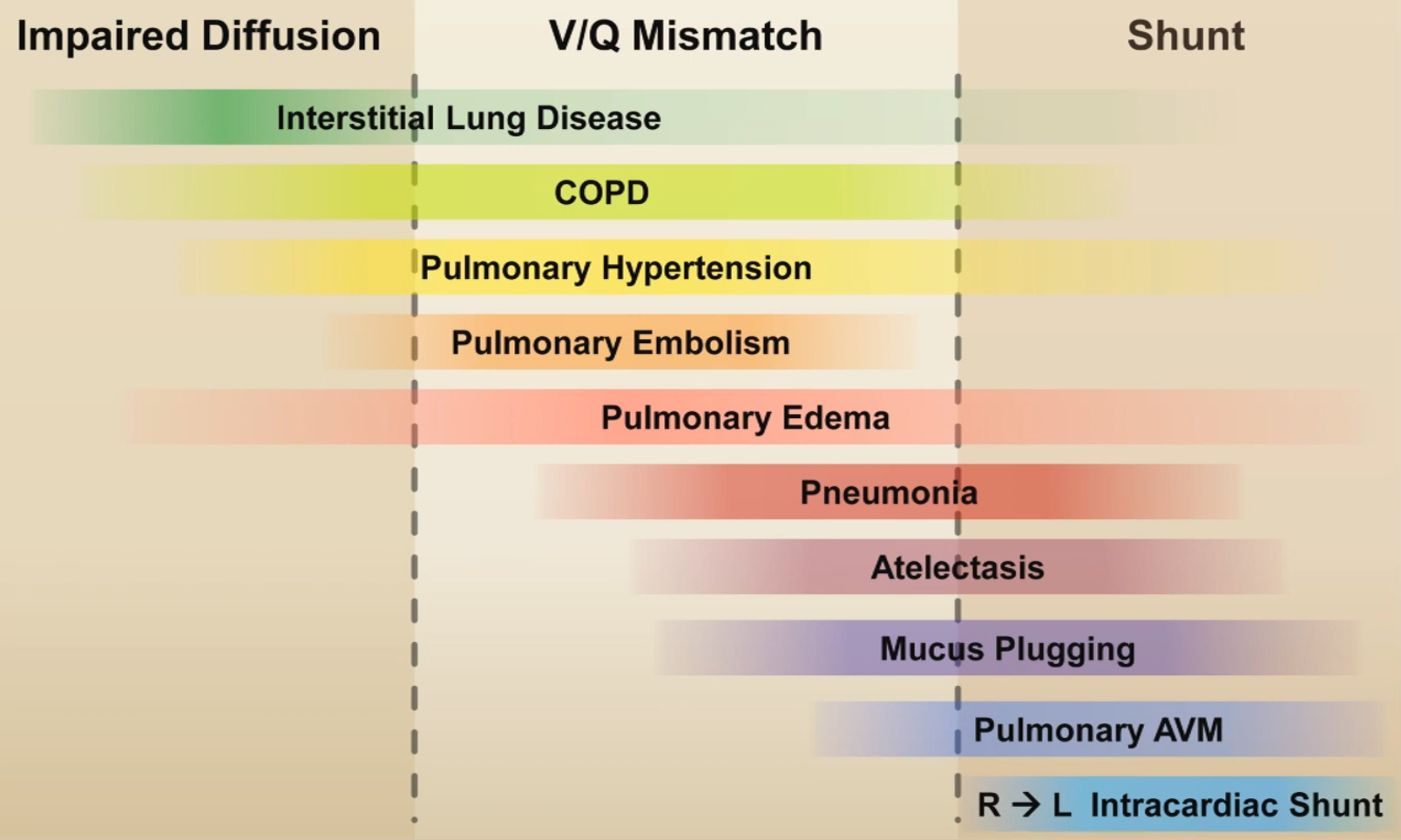
Other Measures of Oxygenation
- Ratio
- Used in ICU setting to measure severity of hypoxaemia
- Normal:
- Abnormal:
- 200-300 mm ⇒ gas exchange abnormal
- <200 mm ⇒ severe abnormality (e.g. more suggestive of ARDS)
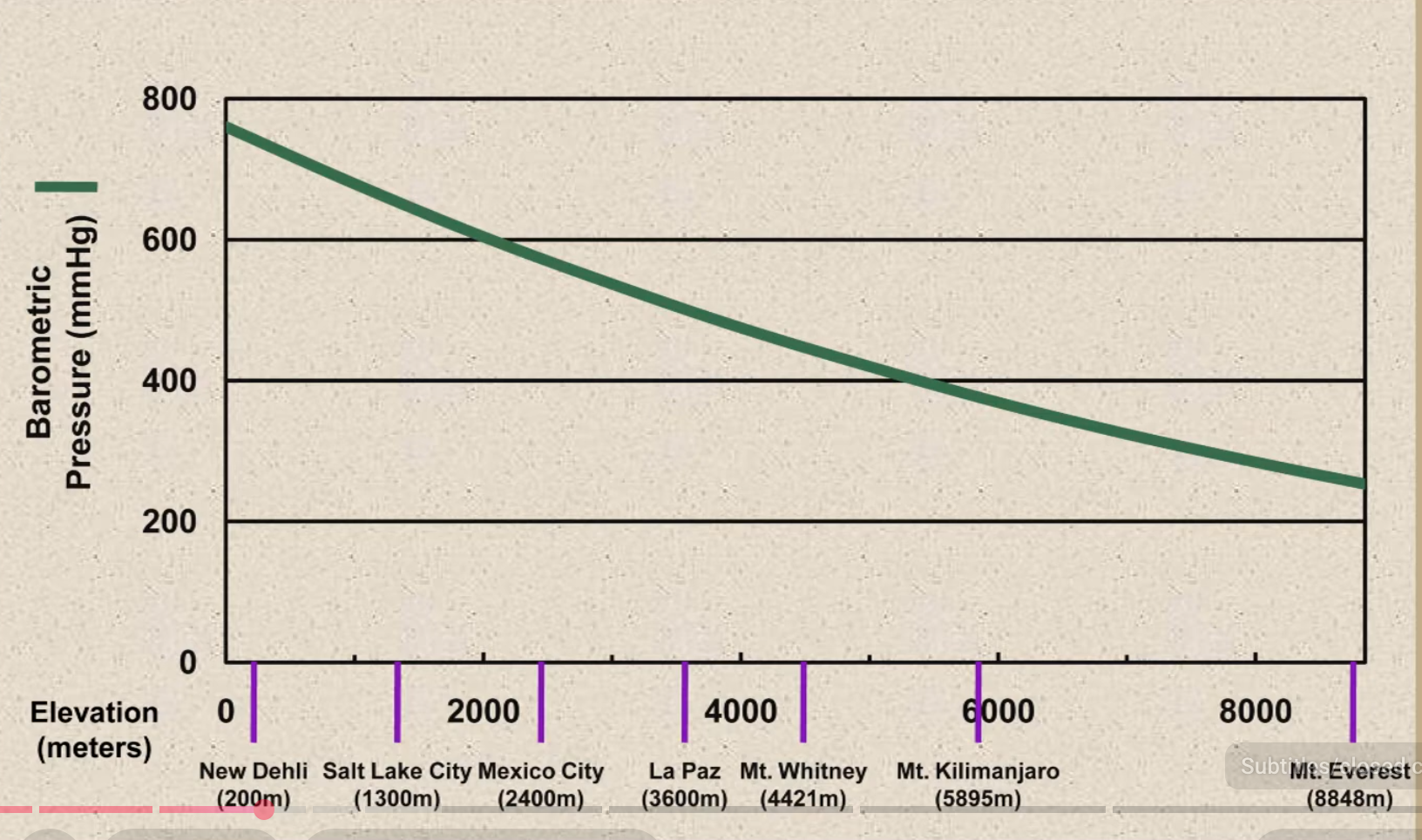
Effect of Altitude
- Higher altitudes will only decrease in the alveolar gas equation (no effect on or )
- usually decreases at high altitudes due to hypoxia-driven hyperventilation
VBG Vs ABG
- While VBGs are not useful for oxygenation status, they can probably substitute for ABGs in most analyses of acid base
- Treger, R., Pirouz, S., Kamangar, N., Corry, D., 2010. Agreement between Central Venous and Arterial Blood Gas Measurements in the Intensive Care Unit. Clin J Am Soc Nephrol 5, 390–394. https://doi.org/10.2215/CJN.00330109
Effect of Temperature on ABG
- Higher temperature → more gas dissolved in blood
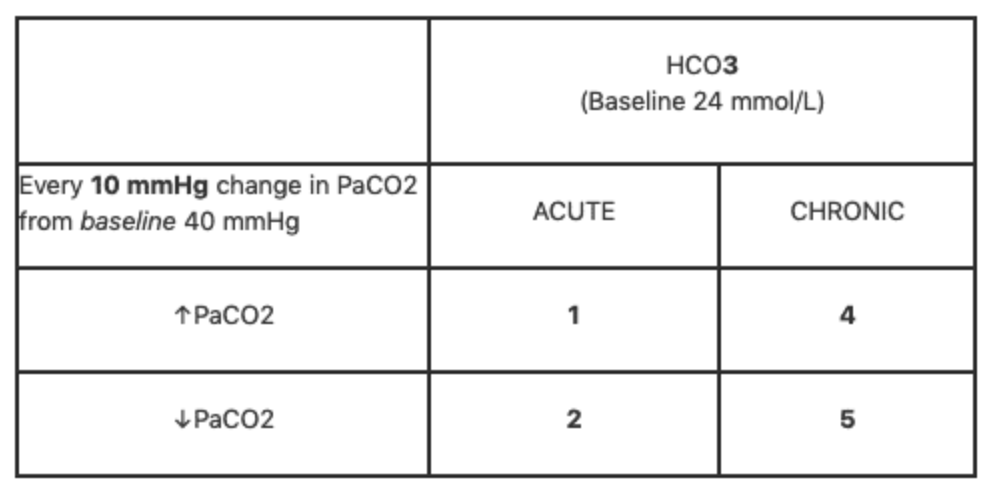
12345 Rule
- Respiratory acidosis: For every 10 mmHg rise in PaCO2, there should be 1 increase in for acute and 4 increase in chronic
- Respiratory alkalosis: For every 10mmHg rise in PaCO2, there should be 2 decrease in for acute and 5 decrease in chronic
| Acute | Chronic | |
|---|---|---|
| Respiratory Acidosis | ↑ | ↑ by 4 |
| Respiratory Alkalosis | ↓ by 2 | ↓ by 5 |
Anion Gap
- Given by the formula:
- The normal value varies between machine and technique used (4-12 if measured by ion selective electrode or 8-16 if measured by flame photometry)
- If AG >30 mmol/L then metabolic acidosis is invariably present
- Importantly albumin is the major unmeasured anion and contributes most to the anion gap value, so in hypoalbuminaemia patients may appear as a normal anion gap acidosis but actually have a high anion gap acidosis
- Additionally, you can correct for phosphate as well because hypophosphateaemia also lowers the expected anion gap
- High anion gap metabolic acidosis :: Accumulation of organic acids or impaired excretion
- Normal anion gap metabolic acidosis :: Loss of from ECF See: https://litfl.com/anion-gap/
EXAMPLE
Question A 48 y/o alcoholic man is found unconscious in his apartment, soiled with vomit. He was last seen leaving a party 6 hrs prior. ABG: pH 7.17 / 65 / 22 / Na: 136 / Cl 98 / Albumin 16 What disorders are present? Solution
- pH suggestive of acidaemia
- elevated suggestive of respiratory acidosis
- Check compensation. should increase by 1 for every 10 ⇒ ⇒ lowest allowed value is 23.5, therefore additional metabolic acidosis present
- Calculate anion gap: . ⇒ high anion gap metabolic acidosis
- Calculate delta ratio: which is higher than expected irrespective of the aetiology so there is also a metabolic alkalosis present
- Respiratory acidosis (from intoxication from central respiratory depression) + high anion gap metabolic acidosis (from alcoholic ketoacidosis) + metabolic alkalosis (from vomiting)
Corrected Values
- for glucose
- In patients with marked hyperglycaemia, the elevated serum glucose raises the serum tonicity (as the glucose cannot enter cells) which pulls water out of cells and expands the extracellular water compartment – thereby lowering the concentration of sodium
- The corrected serum sodium can be calculated, which represents what the serum sodium concentration would be if the glucose level was reduced back to normal
- If the corrected sodium is in a normal range – the patient does not have a concurrent hypotonic hyponatraemia
- for pH (acidaemia causes hyperkalaemia)
- For each 0.1 pH drop below 7.4, rises by 0.6 mmol/L
- e.g. For a patient with a of 4 mmol/L at pH 7.2, the corrected is 2.8, hence the patient is hypokalaemic and should be monitored and replaced as pH is corrected
- This occurs because of the exchange of ions for ions across the cell membrane which may cause the appearance of hyperkalaemia on serum potassium levels but the patient is actually net hypokalaemic
- Correcting for potassium in alkalaemia (i.e. alkalaemia causing hypokalaemia) is not often done as the change is quantitatively smaller
- Correcting for albumin, however evidence demonstrates that formulas actually perform worse than uncorrected calcium levels
Easily Figuring Out Direction of Compensation
- One must just look at the Henderson-Hasselbach equation to determine that for the pH to remain the same, the must move in the same direction as and vice versa
Sources
- LITFL
- The A-a Gradient (ABG Interpretation - Lesson 16) - YouTube
- Derranged physiology
- Horowitz Index for Lung Function (P/F Ratio)
- Hypoxia & Hypoxemia — ICU One Pager
Useful resources
Footnotes
-
This is the approach for the ‘Boston’ approach or standard approach ↩
-
In July 2019, Jeremy Cohen (the CICM Chair of the Second Part Exam) issued a decree via the official college newsletter, stating that “unless otherwise specified in the question stem, anion gap calculations should be made without inclusion of potassium or correction to albumin.” ↩
-
Although apparently the evidence for this was from Figge in 1998 and is flawed (see: Mythbusting: Correcting the anion gap for albumin is not helpful) ↩
-
With the exception being patients with extreme diets (e.g. severe anorexia) ↩
-
The normal A-a gradient also increases with higher and may be >100 mmHg in an elderly patient on 100% ↩