- Also see ABG Interpretation
Oxygen Transport
- Oxygen is carried within the circulation bound to Hb (98%) and dissolved in plasma (2%)
- Very little is stored in the body ⇒ apnoea rapidly leads to hypoxia
Red Blood Cells
- Do not contain a nucleus or mitochondria ⇒ entirely dependent on glucose and the glycolytic pathway for energy
Haemoglobin
- HbA has two -chains and two -chains each of which have an iron-containing porphyring ring with iron in the ferrous state ()
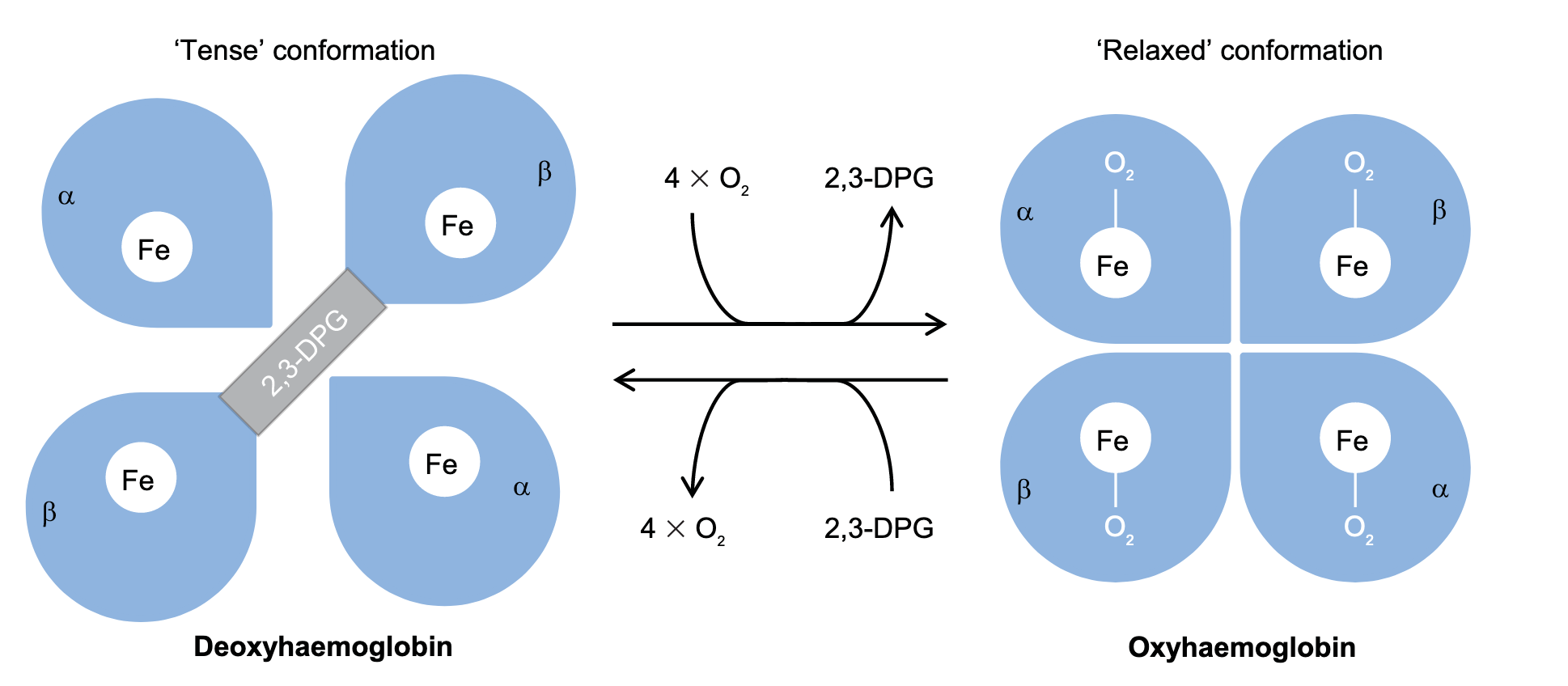
- Haemoglobin has a preference to be either fully saturated or desaturated due to cooperativity
- Cooperative binding is the increase in affinity of Hb with each successive binding
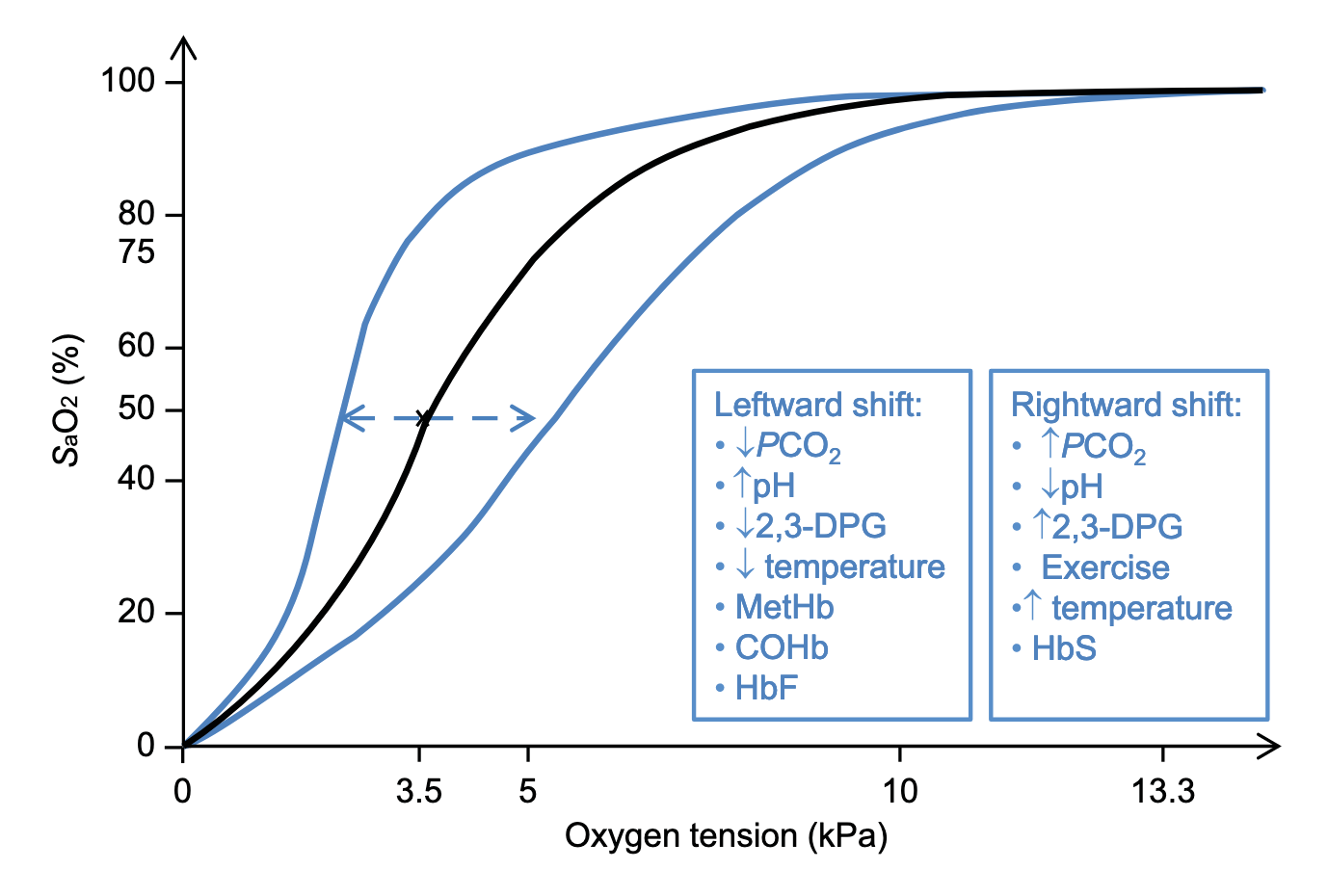
- Rightward shift (↑ offloading) of the oxyhaemoglobin dissociation curve is caused by (mnemonic: CADETS):
- Increased
- Acidosis
- Increased 2,3-dihosphoglycerate
- Exercise
- Increased temperature
- The presence of sickle haemoglobin (HbS) in sickle cell disease
Types of Hb
- Physiological
- : normal most common form ()
- : other adult form accounting for 2-3% ()
- : normal variant during feotal life and has a higher affinity for ()
- Pathological
- : in sickle cell disease which has an abnormal -globin subunit
- : Methaemoglobinaemia is where the ferrous iron within the Hb molecule is oxidised to ferric iron (); cannot bind to , so cannot participate in transport
- Low level of is maintained by:
- Glutathione/nicotinamide adenine dinucleotide phosphate (NADPH) system where oxidising agents are reduced by glutathione before being able to oxidise ferrous iron; the pentose phosphate pathway is integral for this as it supplies NADPH to return glutathione back to its reduced form
- reductase/nicotinamide adenine dinucleotide (NADH) system reduces ferric iron by a reduction system involving reductase
- Methaemoglobinaemia occurs as a result of:
- Oxidising agents overwhelming the glutathione system (e.g. suphonamide antibiotics, nitric oxide and prilocaine)
- Failure of protective reduction systems (e.g. G6PD deficiency)
- Pulse oximeters misread and display values of 85% irrespective of concentration
- Manage with supplemental oxygen in mild cases and methylene blue in severe cases
- Low level of is maintained by:
- : Formed when binds inhaled carbon monoxide molecules
- : Cyanohaemoglobin is formed when is exposed to cyanide ions
Carbon Dioxide Transport
- A typical adult produces at a basal rate of 200mL/min; during exercise it can increase as high as 4000 mL/min
- is transported in circulation in three forms
- Dissolved in plasma
- Bound to Hb and other proteins as carbamino compounds
- When reacts with a terminal amine group within the Hb molecule forming carbaminohaemoglobin
- Deoxyhaemoglobin forms carbamino compounds more readily than oxyhaemoglobin
- Bicarbonate
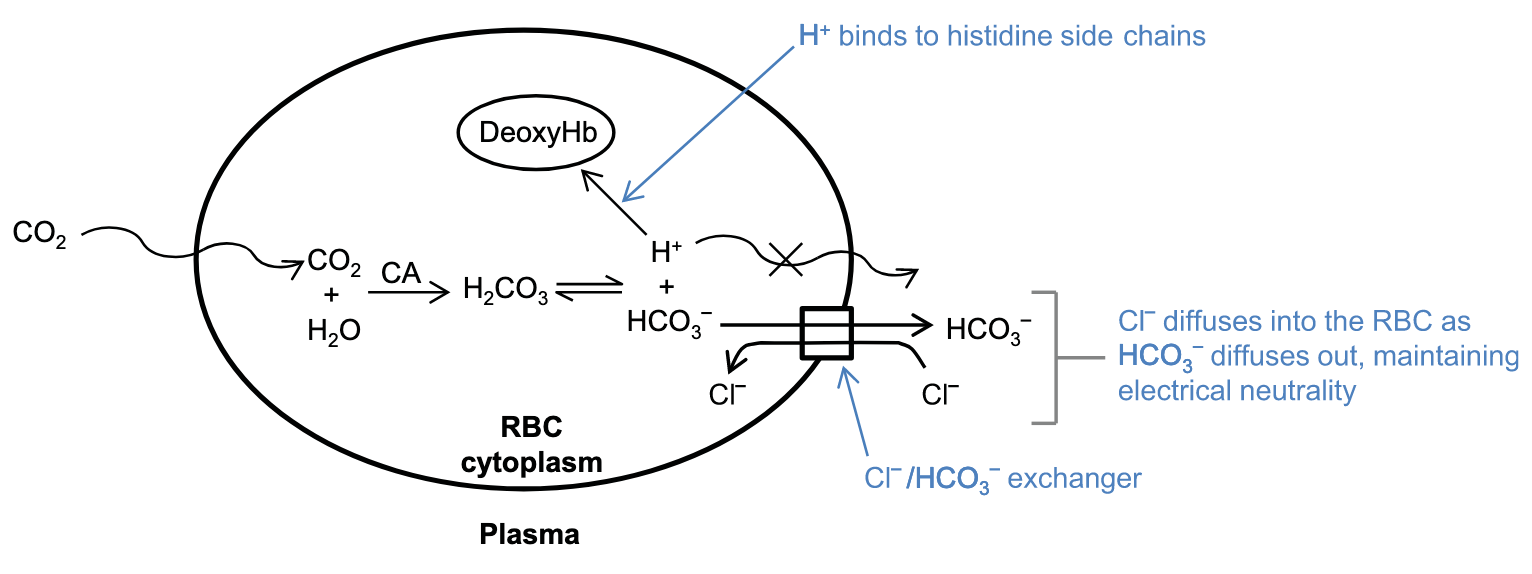
- Carbonic anhydrase catalyses the reaction to form bicarbonic acid
- The cytoplasm of red blood cells contains carbonic anhydrase, but plasma does not contain carbonic anhydrase therefore it can only occur within the RBC
- Almost all dissociates into and
- and water can diffuse directly through the RBC membrane whilst and cannot; forward direction of the reaction is maintained by preventing the build up and via two processes:
- Chloride shift (Hamburger effect): is transported across teh RBC membrane down its electrochemical gradient by the / exchanger
- Binding of to histidine side chains of the haemoglobin molecule reducing intracellular concentration of
- Carbonic anhydrase catalyses the reaction to form bicarbonic acid
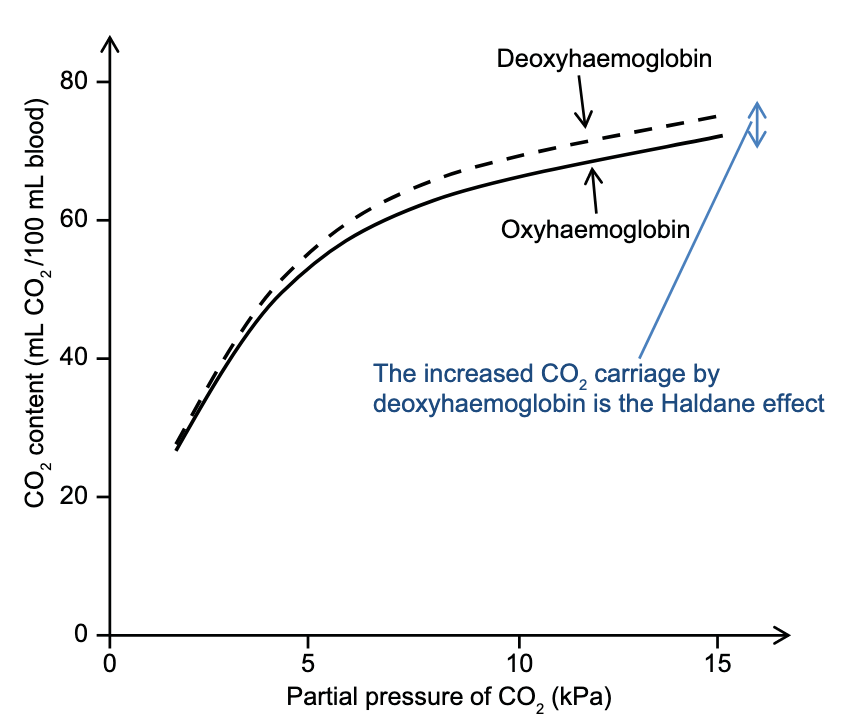
Haldane Effect
- Describes the observation that deoxyhaemoglobin is more effective net carrier of than oxyhaemoglobin
- This is because of two reasons:
- Deoxyhaemoglobin more readily forms carbamino compounds
- Deoxyhaemoglobin is a stronger base than oxyhaemoglobin and more readily accepts allowing rightward shift of the above equilibrium (increasing formation)
- Metabolically active tissues produce and . Through the Bohr effect and its effect on , additional is offloaded to the most metabolically active tissues
- According to the Haldane effect, the newly formed deoxyhaemoglobin is better at binding and carrying than is oxyhaemoglobin
- The metabolic waste products are therefore efficiently transported away from the tissues to the lungs
Carbon Dioxide Dissociation Curve
- Describes the relationship between partial pressure of () and the blood content
- Typical is 5.3 kPa (~39.8 mmHg) and results in a content of about 48 mL/100 mL
- Typical is 6.1 kPa (~45.8 mmHg) and results in a content of about 52 mL/100 mL
- At physiological , the dissociation curve is essentially linear
- Often drawn as two curves representing arterial and venous blood
Importance of Pre-Oxygenation During Intubation
In total, the circulation and lungs contain approximately 2.5 L of immediately available and 1550 mL of . If a healthy patient stops breathing (e.g. on induction of general anaesthesia), basal processes will continue: 250 mL/min of will be consumed and 200 mL/min of will be produced. Therefore: will increase by 0.4–0.8 kPa/min. will fall. The rate of fall is complicated, involving factors such as Hb concentration and total blood volume. Typically, falls to 70% ( 5.0 kPa) after 2 min. However, if the patient breathes for sufficient time to completely de-nitrogenate their functional residual capacity prior to the period of apnoea, the quantity of stored increases to over 3 L – even after 5 min of apnoea, will remain at 100%. Basal metabolic processes will continue and after 5 min the will approach 10 kPa.
Lung Mechanics
Definitions
- Where:
- is the minute ventilation
- is the tidal volume
- Where:
- is the alveolar ventilation
- is teh physiologic dead space
- Where
- is airflow
- is pressure gradient
- is airway resistance
Source
Chambers, D., Huang, C., Matthews, G., 2019. Basic Physiology for Anaesthetists, 2nd ed. Cambridge University Press. https://doi.org/10.1017/9781108565011